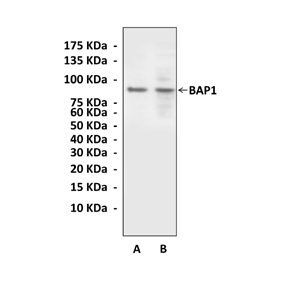
Anti-BAP1: Mouse BRCA1-Associated Protein 1 Antibody |
 |
BACKGROUND BRCA1-associated protein 1 (BAP1) is a nuclear-residing ubiquitin COOH-terminal hydrolase (UCH), a subfamily of deubiquitinating enzymes (DUB), which was originally identified by the yeast two-hybrid technique as a protein that interacted with the RING finger domain of BRCA1 and enhanced BRCA1-mediated cell growth suppression.1 DUBs are proteases that catalyze the hydrolysis of the isopeptide linkage that joins the COOH-terminal glycine of ubiquitin and a lysine side chain on the target polypeptide, resulting in removal of single ubiquitin moieties from ubiquitin chains or cleavage of the isopeptide bond between ubiquitin and the substrate protein. The DUB enzymes are classified into five categories: UCH, the ubiquitin-specific processing proteases (UBP or USP), Jab1/Pad1/MPN domain containing metalloproteinases (JAMM), Otu domain ubiquitin-aldehyde binding proteins (OTU), and Ataxin-3/Josephin domain containing proteins (Ataxin-3/Josephin). Whereas the USP family includes at least 58 members, the UCH family consists of only 4 members: UCH-L1, UCH-L3, UCH-L5/UCH37, and BAP1. Whereas UCH enzymes are generally small, containing only a proteolytic core domain, BAP1 is a relatively large protein with an NH2-terminal proteolytic core domain and a COOH-terminal domain that contains nuclear localization signals and interacts with the BRCA1 RING finger. However, it was shown that BAP1 was incapable of deubiquitinating autoubiquitinated BRCA1, although it possessed ubiquitin hydrolase activity.2 As known, the breast and ovarian tumor suppressor BRCA1 constitutes a RING heterodimer E3 ligase with BARD1. BAP1 inhibits the E3 ligase activity of BRCA1/BARD1 by binding to BARD1 and perturbing RING heterodimer formation in addition to deubiquitinating BRCA1/BARD1-mediated ubiquitination. Thus, that BRCA1-mediated ubiquitination and BAP1-mediated ubiquitination-suppression may coordinately function in these cellular processes.3 In addition, it was shown that BAP1 itself possesses growth inhibitory activity and functions as a tumor suppressor. Moreover, BAP1 exerts its tumor suppressor functions by affecting the cell cycle; speeding the progression through the G1/S checkpoint and inducing cell death via a process that has characteristics of both apoptosis and necrosis. Surprisingly, BAP1-mediated growth suppression is independent of wild-type BRCA1.4 On the other hand, BAP1 also plays positive roles in cell proliferation. BAP1 depletion by RNAi inhibits cell proliferation as does overexpression of a dominant negative mutant of BAP1. Mass spectrometry analyses of copurified proteins revealed that BAP1 is associated with factors involved in chromatin modulation and transcriptional regulation. We show that the interaction with host cell factor-1 (HCF-1), a cell-cycle regulator composed of HCF-1N and HCF-1C, is critical for the BAP1-mediated growth regulation. HCF-1N is modified with Lys-48-linked polyubiquitin chains on its Kelch domain. BAP1 regulates cell proliferation by deubiquitinating HCF-1.5 Furthermore, other studies showed that BAP1 assembles multiprotein complexes containing numerous transcription factors and cofactors, including HCF-1 and the transcription factor Yin Yang 1 (YY1). Through its coiled-coil motif, BAP1 directly interacts with the zinc fingers of YY1. Moreover, HCF-1 interacts with the middle region of YY1 encompassing the glycine-lysine-rich domain and is essential for the formation of a ternary complex with YY1 and BAP1 in vivo.6
REFERENCES
1. Jensen, D.E. et al: Oncogene 16:1097–112, 1998
2. Mallery, D.L. et al: EMBO J. 21:6755–62, 2002
3. Nishikawa, H. et al: Cancer Res. 69:111–9, 2009
4. Ventii, K.H. et al: Cancer Res. 68:6953-62, 2008
5. Machida, Y.J. et al: J. Biol. Chem. 284:34179-88, 2009
6. Yu, H. et al: Mol. Cell. Biol. 30:5071-85, 2010
2. Mallery, D.L. et al: EMBO J. 21:6755–62, 2002
3. Nishikawa, H. et al: Cancer Res. 69:111–9, 2009
4. Ventii, K.H. et al: Cancer Res. 68:6953-62, 2008
5. Machida, Y.J. et al: J. Biol. Chem. 284:34179-88, 2009
6. Yu, H. et al: Mol. Cell. Biol. 30:5071-85, 2010
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CA11002 |
Antigen: | Short peptide from human BAP1 sequence. |
Isotype: | Mouse IgG |
Species & predicted species cross- reactivity ( ): | Human, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 81 kDa |
Specificity/Sensitivity: | Detects endogenous levels of BAP1 proteins without cross-reactivity with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой