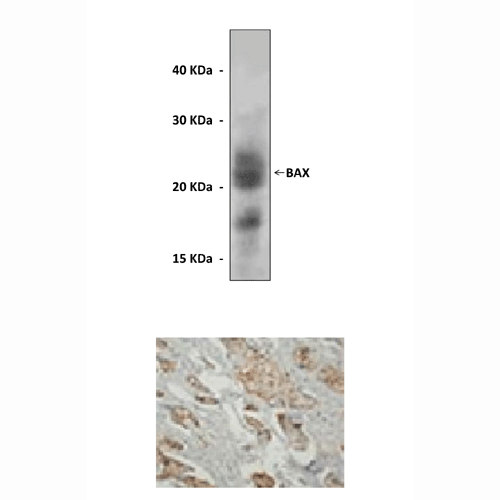
Anti-Bax: Polyclonal Bcl-2 Associated X Protein Antibody |
 |
BACKGROUND Bcl-2 family proteins regulate and contribute to programmed cell death or apoptosis. It is a large protein family and all members contain at least one of four BH (bcl-2 homology) domains. The Bcl-2 family of proteins governs mitochondrial membrane permeability and can be either pro-apoptotic or anti-apoptotic. To date, a total of 25 genes have been identified in the Bcl-2 family. Some of the anti-apoptotic proteins include Bcl-2, Bcl-x, Bcl-XL, Bcl-XS, Bcl-w, BAG, and some of the pro-apoptotic proteins include Bcl-10, Bax, Bak, Bid, Bad, Bim, Bik, and Blk. These proteins have special significance since they can determine if the cell commits to apoptosis or aborts the process. It is thought that the main mechanism of action of the Bcl-2 family of proteins is the regulation of cytochrome c release from the mitochondria via alteration of mitochondrial membrane permeability. The pro-apoptotic group of Bcl-2 proteins can be further sub-divided into the structurally diverse 'BH3' only proteins (e.g. Bid, Noxa, Puma and Bad) and the multidomain proteins that share BH1 to 3 (e.g. Bax and Bak). Most Bcl-2 family members contain a C-terminal transmembrane domain that functions to target these proteins to the outer mitochondrial and other intracellular membranes.1
Bak (a Bcl-2 homologous antagonist/killer) has been cloned as a Bcl-2-related gene.2 Genetic studies show that the multidomain proapoptotic proteins Bak and Bax, which partially complement one another, are required for apoptosis induced by many types of death stimulation. In healthy cells, Bax is distributed in the cytoplasm or loosely attached to membranes, whereas Bak is mainly located on the outer mitochondrial membrane (OMM). Apoptotic signals induce conformation changes in the Bax and Bak proteins, leading to Bax translocation to OMM and the formation of membrane-integrated homo-oligomers of Bax and Bak, which results in the outer mitochondrial membrane permeabilization (OMMP). The antiapoptotic Bcl-2 family members, such as Bcl-2 and Bcl-XL, inhibit Bax and Bak activation, whereas the BH3-only proteins promote it. Most of these BH3-only proteins appear to function as transdominant inhibitors that activate Bax and Bak by binding to and antagonizing antiapoptotic Bcl-2 family proteins, with the exception of tBid and BimS or BimAD, which are capable of inducing OMMP by direct binding to and activation of Bax and Bak. Although BH3-only proteins are essential for mitochondrial apoptosis, the precise mechanism underlying both Bax and Bak activation remains far from clear. Several non-Bcl-2-related proteins, including Bif-1, Ku70, 14-3-3 theta, Humanin, ASC, and p53, have recently been shown to be involved in the regulation of Bax and/or Bak activity.3
REFERENCES
1. Elmore S.: Toxicol. Pathol. 34:495-516, 2007
2. Karbowski,M. et al: J. Cell Sci. 166:1027-39, 2004
3. Takahashi, Y. et al:Mol. Cell. Biol. 25:9369-82, 2005
2. Karbowski,M. et al: J. Cell Sci. 166:1027-39, 2004
3. Takahashi, Y. et al:Mol. Cell. Biol. 25:9369-82, 2005
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CA0315 |
Antigen: | N- terminal sequence of human BAX |
Isotype: | Affinity-purified rabbit polyclonal IgG |
Species & predicted species cross- reactivity ( ): | human, rabbit, rat, mouse |
Applications & Suggested starting dilutions: | WB 1:500 to 1:1000 IP n/d IHC (Paraffin) 1:50 to 1:200 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 21 kDa |
Specificity/Sensitivity: | Reacts specifically with BAX of human, rabbit, mouse & rat origin in Immunohistochemistry cytoplasmic staining) and western blotting, non cross-reactive with BCL-2 and other apoptosis associated protein. |
Storage: | Store at 4° C for frequent use; at -20° C for at least one year. |
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой