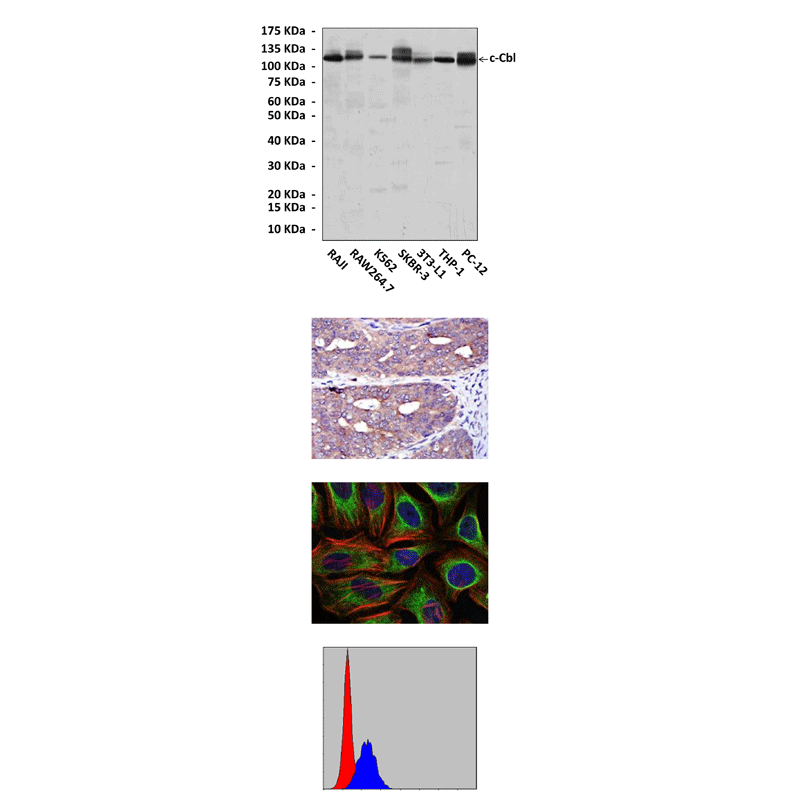
Anti-c-Cbl: Mouse c-Cbl Antibody |
 |
BACKGROUND Cbl has emerged as a novel signal transducing protein downstream of a number of cell surface receptors coupled to tyrosine kinases. Identified as the protein product of the c-Cbl proto-oncogene, the cellular homolog to the transforming gene of a murine retrovirus, Cbl comprises an N-terminal transforming region (Cbl-N), which contains a phosphotyrosine binding (PTB) domain, and a C-terminal modular region (Cbl-C) containing a RING finger motif, a large proline-rich region and a leucine zipper. Deletion of Cbl-C or small deletions N-terminal to the RING finger render Cbl oncogenic, whereas wild type Cbl is non-transforming, even if overexpressed.1 Cbl serves as a substrate of both receptor and non-receptor tyrosine kinases, and binds to adaptor proteins Grb2, Crk and the p85 subunit of PI-3-kinase and mediates the downstream signaling.2 On the other hand, acting as an E3 ubiquitin-protein ligase, which accepts ubiquitin from specific E2 ubiquitin-conjugating enzymes, and then transfers it to substrates promoting their degradation by the proteasome, Cbl also regulates receptor protein-tyrosine kinase ubiquitination in a manner dependent upon its variant SH2 and RING finger domains. Ubiquitination of receptor protein-tyrosine kinases terminates signaling by marking active receptors for degradation.3
REFERENCES
1. Lupher, M.L. Jr. et al: Int. J. Biochem. Cell Biol. 30:439-44, 1998
2. Smit, L. & Borst, J.:Crit. Rev. Oncog. 8:359-79, 1997
3. Thien, C.B.F. & Langdon, W.Y.: Biochem. J. 391:153-166, 2005
2. Smit, L. & Borst, J.:Crit. Rev. Oncog. 8:359-79, 1997
3. Thien, C.B.F. & Langdon, W.Y.: Biochem. J. 391:153-166, 2005
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10401 |
Antigen: | Raised against recombinant human c-Cbl fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP 1:50 IHC 1:50 - 1:200 ICC 1:50 - 1:200 FACS 1:50 - 1:200 |
Predicted Molecular Weight of protein: | 120 kDa |
Specificity/Sensitivity: | Detects c-Cbl proteins in various cell lysate. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой