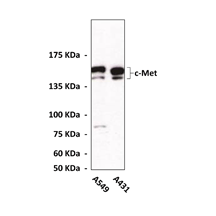
Anti-c-Met: Polyclonal c-Met Antibody |
 |
BACKGROUND The c-Met proto-oncogene, widely expressed in mammalian tissues, encodes a transmembrane receptor with tyrosine kinase activity. The c-Met receptor tyrosine kinase and its ligand HGF (hepatocyte growth factor), also known as scatter factor (SF), have been shown to be involved in angiogenesis, cellular motility, growth, invasion, and differentiation. The full receptor protein is a 190-kDa heterodimer made of a 50-kDa extracellular alpha-subunit linked by a disulfide bridge to a 145-kDa transmembraneous catalytic beta-subunit.1 Multiple signaling pathways have been associated with the biological responses mediated by c-Met activation. In response to HGF, phosphorylated tyrosine residues of c-met act as a binding site for Src Homology (SH2) domain-containing intracellular proteins including Gab-1, Grb2 etc.2, which led to downstream activation of Ras, Rac, phosphoinositide 3-kinase (PI3-kinase) and mitogen-activated protein kinase (MAPK).2 It was demonstrated that c-Met signaling also contributes to oncogenesis and tumor progression in several human cancers and promotes aggressive cellular invasiveness that is strongly linked to tumor metastasis. Thus, c-Met is a potentially therapeutic target for cancer treatment.3
REFERENCES
1. Abounader, A et al: J. Neurochem. 76 : 1497, 2001.
2. Sachs, M. et al: J. Cell. Biol. 150:1375, 2000.
3. Perruzi, B. & Bottaro, D.: Clin. Can. Res. 12: 3657, 2006
2. Sachs, M. et al: J. Cell. Biol. 150:1375, 2000.
3. Perruzi, B. & Bottaro, D.: Clin. Can. Res. 12: 3657, 2006
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CB2706 |
Antigen: | E. coli-expressed cytoplasmic carboxyl domain of human c-Met proteins. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 145 kDa |
Specificity/Sensitivity: | Detects endogenous levels of c-Met beta subunits |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой