Anti-DDR2: Mouse Discoidin Domain Receptor 2 Antibody |
 |
BACKGROUND Discoidin domain receptors (DDRs) are a subfamily of transmembrane collagen-binding receptor tyrosine kinases (RTK). DDRs are distinguished from other RTKs by a discoidin domain in their extracellular region, which functions as a lectin in the slime mold Dictyostelium discoideum. There are two genes that code for DDRs in humans, DDR1 and DDR2. Each DDR contains an extracellular region containing the ligand-binding discoidin domain, a stalk region, and a single transmembrane region, as well as a cytoplasmic domain consisting of a juxtamembrane region and a tyrosine kinase domain.1 DDRs signal in response to collagens rather than soluble peptide growth factors, and they display a relatively slow onset of phosphorylation, occurring in hours rather than minutes. The triple helical structure of collagen is required for activation of both DDR1 and DDR2. However, DDR1 expression is confined to epithelial tissues where it is activated equally by collagen types I, IV, and V. In contrast, DDR2 is found in mesenchymal cells and is activated primarily by collagen type I, and to a lesser extent, by collagen types II, III, and V. DDR2 is also present at high levels in stromal cells surrounding DDR1-expressing epithelial human tumor cells.2 Silencing the expression of DDRs diminish the key functional characteristics of tumor cells to progress and metastasize. To date, the biological outcomes of DDR activation have not been well characterized. DDR1 signaling is also essential for granule cell axon extension in the developing cerebellum. Prolonged activation of DDR2 has been associated with increased MMPs activity in the human fibrosarcoma cell line HT-1080.3 Recently, it was demonstrated that the growth inhibitory activity of fibrillar collagen on tumor cells is the result of activation of DDR2 tyrosine kinase activity, which led to growth arrest of tumor cells in the G0/G1 phase of the cell cycle.4
REFERENCES
1. Abdulhussein, R. et al: J. Biol. Chem. 283:12026-12033, 2008
2. Alves, L. et al: Oncogene 10:609-18, 1995
3. Vogel, W. et al: Mol. Cell 1:13-23, 1997
4. Wall, S.J. et al: J. Biol. Chem. 280:40187-94, 2005
2. Alves, L. et al: Oncogene 10:609-18, 1995
3. Vogel, W. et al: Mol. Cell 1:13-23, 1997
4. Wall, S.J. et al: J. Biol. Chem. 280:40187-94, 2005
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP11068 |
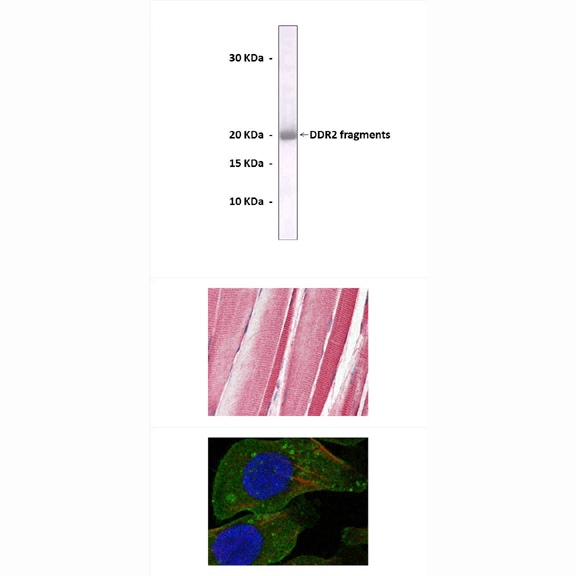
Antigen: | Purified recombinant human DDR2 fragments expressed in E. coli. |
Isotype: | Mouse IgG2a |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP 1:50 IHC 1:100 ICC 1:100 FACS n/d |
Predicted Molecular Weight of protein: | 120 kDa |
Specificity/Sensitivity: | Detects endogenous DDR2 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой