Anti-DLK1: Mouse DLK1 Antibody |
 |
BACKGROUND The paternally expressed Delta-like 1 homolog (Dlk1) gene (Entrez Gene ID 13386) lies within the imprinted Dlk1 – Dio3 gene cluster on distal mouse chromosome 12 and encodes a type I transmembrane epidermal growth factor (EGF)-like protein. The extracellular domain of Dlk1 can also be cleaved to generate a soluble form, called fetal antigen 1, which circulates as an abundant growth factor during development. Another splice variant lacks the proteolytic cleavage site and remains constitutively membrane bound (Dlk1-C2). This membrane-bound form is most common in postnatal skeletal muscle. DLK1 is similar to other members of the Notch/Delta/Serrate family. It is expressed in animals from birds to mammals extensively in immature cells, and is down-regulated during fetal development, which implicates that DLK1 plays an important role in fetal development and growth. However, DLK1 is only detected in selected adult tissues and several tumor cells including neuroblastoma (NB), gliomas, small-cell lung carcinoma, and leukemia. Previous studies have demonstrated that exogenous DLK1 increased NB cell clonogenic growth, which is a self-renewal characteristics of cancer stem cells, and regulated growth of pre-B cells and thymocytes. Those results suggest that DLK1 performs an important role in stem/progenitor and cancer cell growth. Accumulating evidence further indicates that Dlk1 is an important regulator of not only proliferation and differentiation of embryonic and adult stem cells but also functions to maintain the pluripotency of embryonic stem cells.1
A role of DLK1 in adipogenesis has been well documented. Recently, DLK1 was shown to regulate fate of myogenic cells and human skeletal stem cells. Transcript and protein levels of Dlk1 are highest in developing fetal muscle and taper off quickly after birth. It was shown that the DLK1 protein was highly abundant in the endomycium surrounding individual myofibers and the perimycium separating fiber bundles of hypertrophied callipyge muscles. The increased Dlk1 expression has been associated with muscle hypertrophy in animal models. It was demonstrated that Dlk1 expressed by nascent or regenerating myofibers non-cell autonomously promotes the differentiation of their neighbor satellite cells and therefore leads to muscle hypertrophy.2 A significant role of DLK1 in maintaining proper organism function was demonstrated by generating DLK1 knockout-mice, which exhibited accelerated obesity, growth disorders and skeletal malformation. In addition, it was demonstrated that an elevated DLK1 level provided a survival advantage for NB cells in a poor microenvironment characterized by serum and glucose deprivation. Increasing severity of nutrition deprivation and knock-down of DLK1 caused greater apoptotic death of NB cells. Thus, DLK1 could provide a novel therapeutic strategy for regulating apoptosis in NB cells.3
A role of DLK1 in adipogenesis has been well documented. Recently, DLK1 was shown to regulate fate of myogenic cells and human skeletal stem cells. Transcript and protein levels of Dlk1 are highest in developing fetal muscle and taper off quickly after birth. It was shown that the DLK1 protein was highly abundant in the endomycium surrounding individual myofibers and the perimycium separating fiber bundles of hypertrophied callipyge muscles. The increased Dlk1 expression has been associated with muscle hypertrophy in animal models. It was demonstrated that Dlk1 expressed by nascent or regenerating myofibers non-cell autonomously promotes the differentiation of their neighbor satellite cells and therefore leads to muscle hypertrophy.2 A significant role of DLK1 in maintaining proper organism function was demonstrated by generating DLK1 knockout-mice, which exhibited accelerated obesity, growth disorders and skeletal malformation. In addition, it was demonstrated that an elevated DLK1 level provided a survival advantage for NB cells in a poor microenvironment characterized by serum and glucose deprivation. Increasing severity of nutrition deprivation and knock-down of DLK1 caused greater apoptotic death of NB cells. Thus, DLK1 could provide a novel therapeutic strategy for regulating apoptosis in NB cells.3
REFERENCES
1. Liu, L. et al: J. Biol. Chem. 285:19483–90, 2010
2. Waddell,J.N. et al: PLoS ONE 5: e15055, 2010
3. Kim, Y.: Nutr. Res. Pract. 4:455–61, 2010
2. Waddell,J.N. et al: PLoS ONE 5: e15055, 2010
3. Kim, Y.: Nutr. Res. Pract. 4:455–61, 2010
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10427 |
Antigen: | Raised against recombinant human DLK1 fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse |
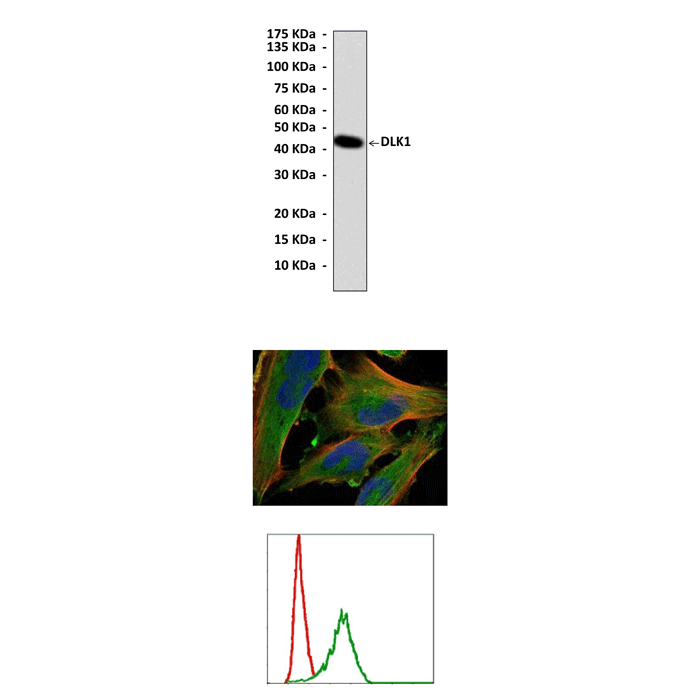
Applications & Suggested starting dilutions:* | WB 1:1000 IP 1:50 IHC n/d ICC 1:50 - 1:200 FACS 1:50 - 1:200 |
Predicted Molecular Weight of protein: | 42 kDa |
Specificity/Sensitivity: | Detects endogenous DLK1 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой