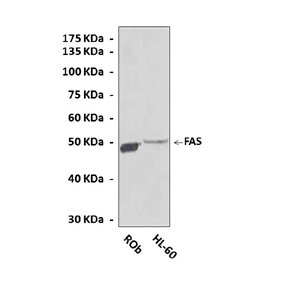
Anti-FAS: Polyclonal FAS Antibody |
 |
BACKGROUND FAS, known also as CD95, Apo-1, and TNFRSf6, belongs to the subgroup of the tumor necrosis factor receptor (TNF-R) family that contains an intracellular death domain (DD) and triggers apoptosis. Its physiological ligand FASL is a member of the TNF cytokine family.1 Fas forms the death inducing signalling complex (DISC) upon ligand binding.2 Membrane-anchored Fas ligand trimer on the surface of an adjacent cell causes trimerization of Fas receptor. Upon ensuing death domain (DD) aggregation, the receptor complex is internalized via the cellular endosomal machinery. This allows the adaptor molecule FADD to bind the death domain of Fas through its own death domain. FADD also contains a death effector domain (DED) near its amino terminus, which facilitates binding to the DED of FADD-like ICE (FLICE), more commonly referred to as caspase-8.3 FLICE can then self-activate through proteolytic cleavage into p10 and p18 subunits, two each of which form the active heterotetramer enzyme. Active caspase-8 is then released from the DISC into the cytosol, where it cleaves other effector caspases, eventually leading to DNA degradation, membrane blebbing, and other hallmarks of apoptosis.4
REFERENCES
1. Strasser A et al.: Immunity, 30:180-192, 2009.
2. Mundle SD & Raza A: Immunol. Today, 23: 187-194, 2002.
3. Qiu J et al.: J. Neurosci., 22:3504-3511, 2002.
4. Tsuyuki et al.: J Clin Invest., 96: 2924–2931, 1995.
2. Mundle SD & Raza A: Immunol. Today, 23: 187-194, 2002.
3. Qiu J et al.: J. Neurosci., 22:3504-3511, 2002.
4. Tsuyuki et al.: J Clin Invest., 96: 2924–2931, 1995.
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CA1119 |
Antigen: | Peptide containing human FAS C-terminal sequence. |
Isotype: | Rabbit Polyclonal |
Species & predicted species cross- reactivity ( ): | Human, Rat, Mouse |
Applications & Suggested starting dilutions: | WB 1:1000 IP 1:50 IHC (Paraffin) 1:100 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 50 kDa |
Specificity/Sensitivity: | Detects endogenous levels of FAS protein in normal primary cell lysates. |
Storage: | Store at 4° C for frequent use; at -20° C for at least one year. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой