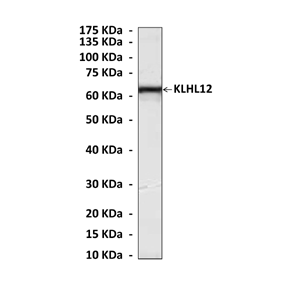
Anti-KLHL12: Mouse KLHL12 Antibody |
 |
BACKGROUND The Kelch-related proteins are a superfamily of proteins conserved in a wide range of organisms, from viruses to mammals. At least 60 Kelch-related proteins have been identified, but their physiological and biochemical functions remain largely uncharacterized. Rearrangement of the actin-based cytoskeleton is regulated by a large number of actin-binding proteins. The kelch-related proteins are believed to be important for the maintenance of the ordered cytoskeleton. The Drosophila Kelch proteins colocalize with actin filaments in a structure called the ring canal, which bridges 15 nurse cells and the oocyte. Drosophila Kelch protein plays an important role in maintaining actin organization during the development of ring canals. The Kelch-related proteins have diverse functions in cell morphology, cell organization, and gene expression, and function in multiprotein complexes through contact sites in their beta-propeller domains. Recently, a new member of the BTB/Kelch repeat family, gigaxonin, was reported to be a pathological target for neurodegenerative disorders in which alterations were found to contain multiple mutations in the Kelch repeats in the neurofilament network. Alterations and mutations of these proteins were found in brain tumors and neurodegenerative disorders.1
KLHL12, also known as C3IP1 or hDKIR, has an N-terminal BTB domain and a C-terminal Kelch domain, linked by a central BACK domain (for “BTB and C-terminal Kelch”). KLHL12 was originally identified to be important in forming ringlike structures in mammalian cells, comparable with the function of the Kelch protein in D. melanogaster. It was shown that KLHL12 is a novel interaction partner of the dopamine D4 receptor that functions as an adaptor in a Cullin3-based E3 ubiquitin ligase complex to target the receptor for ubiquitination. KLHL12 not only interacts with both immature, ER-associated and mature, plasma membrane-associated D4 receptors, but also promotes ubiquitination of both receptor subpools. However, KLHL12-mediated receptor ubiquitination does not promote proteasomal degradation of newly synthesized receptors through the ER-associated degradation pathway or lysosomal degradation of mature receptors. In addition, KLHL12 does also interact with beta-arrestin2 but this has no effect on the ubiquitination or localization of beta-arrestin2 nor on the internalization of the D4 receptor.2 Furthermore, it has been demonstrated that KLHL12 is a important regulator of Wnt signaling pathway. Dishevelled (Dvl) is a conserved protein that interprets signals received by Frizzled receptors. It was shown that KLHL12 E3 ubiquitin ligase complex can be recruited to Dvl in a Wnt-dependent manner that promotes its poly-ubiquitination and degradation. Functional analyses demonstrate that regulation of Dvl by this ubiquitin ligase antagonizes the Wnt–ß-catenin pathway in cultured cells, as well as in Xenopus and zebrafish embryos. Considered with evidence that the distinct Cullin-1 based SCFbeta-TrCPcomplex regulates beta-catenin stability, thus, there are two distinct ubiquitin ligase complexes regulating the Wnt–ß-catenin pathway.3 Overexpression of Dvl is frequently associated with tumors. Therefore, the Dvl protein level must be tightly controlled to sustain Wnt signaling without causing tumors. KLHL12 targets Dvl for ubiquitination and degradation, suggesting its potential importance in avoiding aberrant Dvl overexpression. It was further shown that nucleoredoxin (NRX) determines the Dvl protein level, and NRX expels KLHL12 from Dvl and inhibits ubiquitination. These findings reveal an unexpected function of NRX, retaining a pool of inactive Dvl for robust activation of Wnt/beta-catenin signaling upon Wnt stimulation.4 Finally KLHL12 was identified as a novel autoantigens in Sjögren's syndrome (SjS).5
KLHL12, also known as C3IP1 or hDKIR, has an N-terminal BTB domain and a C-terminal Kelch domain, linked by a central BACK domain (for “BTB and C-terminal Kelch”). KLHL12 was originally identified to be important in forming ringlike structures in mammalian cells, comparable with the function of the Kelch protein in D. melanogaster. It was shown that KLHL12 is a novel interaction partner of the dopamine D4 receptor that functions as an adaptor in a Cullin3-based E3 ubiquitin ligase complex to target the receptor for ubiquitination. KLHL12 not only interacts with both immature, ER-associated and mature, plasma membrane-associated D4 receptors, but also promotes ubiquitination of both receptor subpools. However, KLHL12-mediated receptor ubiquitination does not promote proteasomal degradation of newly synthesized receptors through the ER-associated degradation pathway or lysosomal degradation of mature receptors. In addition, KLHL12 does also interact with beta-arrestin2 but this has no effect on the ubiquitination or localization of beta-arrestin2 nor on the internalization of the D4 receptor.2 Furthermore, it has been demonstrated that KLHL12 is a important regulator of Wnt signaling pathway. Dishevelled (Dvl) is a conserved protein that interprets signals received by Frizzled receptors. It was shown that KLHL12 E3 ubiquitin ligase complex can be recruited to Dvl in a Wnt-dependent manner that promotes its poly-ubiquitination and degradation. Functional analyses demonstrate that regulation of Dvl by this ubiquitin ligase antagonizes the Wnt–ß-catenin pathway in cultured cells, as well as in Xenopus and zebrafish embryos. Considered with evidence that the distinct Cullin-1 based SCFbeta-TrCPcomplex regulates beta-catenin stability, thus, there are two distinct ubiquitin ligase complexes regulating the Wnt–ß-catenin pathway.3 Overexpression of Dvl is frequently associated with tumors. Therefore, the Dvl protein level must be tightly controlled to sustain Wnt signaling without causing tumors. KLHL12 targets Dvl for ubiquitination and degradation, suggesting its potential importance in avoiding aberrant Dvl overexpression. It was further shown that nucleoredoxin (NRX) determines the Dvl protein level, and NRX expels KLHL12 from Dvl and inhibits ubiquitination. These findings reveal an unexpected function of NRX, retaining a pool of inactive Dvl for robust activation of Wnt/beta-catenin signaling upon Wnt stimulation.4 Finally KLHL12 was identified as a novel autoantigens in Sjögren's syndrome (SjS).5
REFERENCES
1. Seng, S. et al: Mol. Cell. Biol. 26:8371-84, 2006
2. Rondou, P. et al: Cell Signal. 22: 900-13, 2010
3. Angers, S. et al: Nat. Cell Biol. 8: 348-57, 2006
4. Funato, Y. et al: Curr. Biol. 21: 1945-52, 2010
5. Uchida, K. et al: Immunol. 116:53-63, 2005
2. Rondou, P. et al: Cell Signal. 22: 900-13, 2010
3. Angers, S. et al: Nat. Cell Biol. 8: 348-57, 2006
4. Funato, Y. et al: Curr. Biol. 21: 1945-52, 2010
5. Uchida, K. et al: Immunol. 116:53-63, 2005
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10337 |
Antigen: | Raised against recombinant human KLHL12 fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 63 kDa |
Specificity/Sensitivity: | Detects endogenous KLHL12 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой