Anti-LMNA: Mouse LMNA Antibody |
 |
BACKGROUND The LMNA gene encodes nuclear lamin A and nuclear lamin C, intermediate filament proteins that are components of the nuclear lamina.1 These A-type lamins are expressed in virtually all differentiated somatic cells. Lamin A (664 amino acids) and lamin C (574 amino acids) arise via alternative splicing of pre-mRNA encoded by exon 10. Lamins A and C are identical for the first 566 amino acids. Lamin A is synthesized as a precursor, prelamin A, which has 98 unique C-terminal amino acids. Prelamin A protein has the amino acids CSIM (Cys-Ser-Ile-Met) at its C-terminus, which triggers a series of enzymatic reactions that lead to farnesylation and carboxylmethylation of the cysteine and endoproteolytic cleavage of the SIM peptide. The farnesylated prelamin A is then recognized by the endoprotease ZMSTE24, which cleaves 15 amino acids up from the cysteine residue at the C-terminus to yield lamin A. By contrast, lamin C has six unique C-terminal amino acids and is not post-translationally modified by farnesylation. Two other genes in the mammalian genome, LMNB1 and LMNB2, encode lamins B1 and B2, respectively. Lamins A and C are widely expressed in the majority of differentiated somatic cells but are lacking from early embryos and from some undifferentiated cells, whereas lamins B1 and B2 are expressed in all or most somatic cells. There are little data and no systemic studies on the differences in the relative amount of lamins A, C, B1 and B2 expression. Lamins are intermediate filament proteins. Like all intermediate filament proteins, lamins contain a highly conserved α-helical core of approximately 350 amino acid residues flanked by globular N-terminal head and C-terminal tail domains. Lamins polymerize within the nucleus to form the nuclear lamina, a meshwork of intermediate filaments. The nuclear lamina is attached to the inner nuclear membrane by the binding of lamins to integral proteins of the membrane, among other factors. One function of the lamina is to provide structural support to the nucleus, and many have argued that defects in this function can lead to disease. Indeed, deficiency of A-type lamins leads to defective nuclear mechanics and defective mechanotransduction in cultured fibroblasts. Nuclear lamins have also been implicated in processes such as chromatin organization, gene regulation, DNA replication and RNA splicing.2 Mutations in the LMNA gene cause a diverse range of diseases, called laminopathies, that selectively affect different tissues and organ systems. The most prevalent laminopathy is cardiomyopathy with or without different types of skeletal muscular dystrophy. LMNA cardiomyopathy has an aggressive clinical course with higher rates of deadly arrhythmias and heart failure than most other heart diseases.3
REFERENCES
1. Shackleton, S. et al: Nature Genet. 24:153-6, 2000
2. Dechat, T. et al:Genes Dev. 22:832-53, 2008
3. Vytopil, M. et al: J. Med. Genet. 40:e132, 2003
2. Dechat, T. et al:Genes Dev. 22:832-53, 2008
3. Vytopil, M. et al: J. Med. Genet. 40:e132, 2003
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10406 |
Antigen: | Raised against recombinant human LMNA fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:50 - 1:200 ICC n/d FACS n/d |
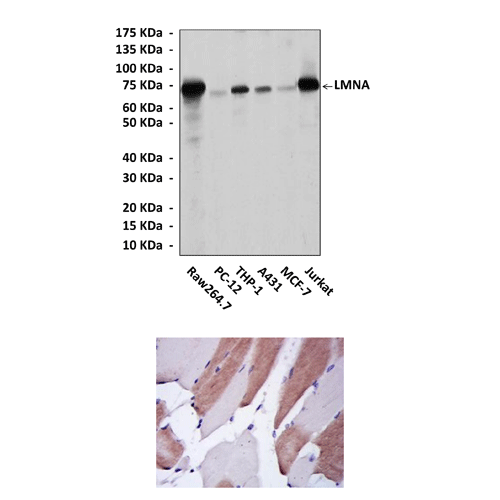
Predicted Molecular Weight of protein: | 74 kDa |
Specificity/Sensitivity: | Detects LMNA proteins in various cell lysate. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой