Anti-LRP4: Rabbit LRP4 Antibody |
 |
BACKGROUND The low-density lipoprotein (LDL) receptor family is a large evolutionarily conserved group of transmembrane proteins. Physiological functions for this ancient gene family include the endocytosis of a large number of macromolecules, including lipoproteins, proteases and protease inhibitors, as well as functions as direct signal transducers or modulators of several fundamental signal transduction pathways, including BMP, TGFβ, PDGF, reelin and canonical Wnt signaling. For example, LDL receptor-related protein 1 (LRP1) is involved in the modulation and integration of PDGF and TGFβ signals in smooth muscle cells of the vascular wall, Apoer2 (LRP8) and its partner Vldlr controls brain development and synaptic transmission through their common signalling ligand Reelin, and Lrp5 and Lrp6 function as co-receptors in the Wnt signalling cascade. Canonical Wnt/β-catenin signalling mediated by LRP5 and LRP6 plays a central role in mammalian bone density regulation.1
LRP4 (also called Megf7) belongs to the LDL receptor family. LRP4 is expressed in various organs, as well as in bone. Mice bearing functional LRP4 null mutations die perinatally due to a failure of forming neuromuscular junctions. Indeed, LRP4 mutant mice display defects in presynaptic and postsynaptic differentiation that are strikingly similar to those found in MuSK mutant mice. Moreover, LRP4 is expressed preferentially by muscle synaptic nuclei, raising the possibility that Lrp4 plays a role in the Agrin/MuSK signaling pathway. It was shown that LRP4 selectively binds neural isoforms of Agrin and is required for Agrin to stimulate MuSK phosphorylation. Moreover, LRP4 self-associates, forms a complex with MuSK, and can reconstitute Agrin-stimulated MuSK phosphorylation in nonmuscle cells that express LRP4 and MuSK. Thus, LRP4 is the receptor for Agrin and has a critical role in activating MuSK and stimulating neuromuscular synapse formation.2 In addition, limb development is also abnormal in LRP4 mutant mice. Hypomorphic mutations of the LRP4 gene are compatible with survival and present with a variable degree of skeletal abnormalities, in particular fusion of digits at the hind and fore limbs (polysyndactyly). Moreover, It was shown that LRP4 integrates BMP and Wnt signaling during tooth development by binding the BMP antagonist Wise. The role of LRP4 as an antagonist of canonical Wnt signaling pathway is thought to be mediated in part by a displacement of the homologous LRP5/6 proteins in the co-receptor complex formed by frizzled proteins (fzd) with LRP5/6, which is required to bind Wnt proteins and to transduce the Wnt signal to downstream elements of the canonical cascade. Dickkopf1 (Dkk1) is another soluble inhibitor of Wnt/β-catenin signaling that binds to LRP5 and LRP6. And sclerostin is a potent osteocyte secreted inhibitor of bone formation that directly binds to LRP5 and LRP6. The powerful anti-osteogenic effect of sclerostin is thought to be mediated mainly by inhibition of Wnt-signaling through LRP5/6 within osteoblasts by disrupting the Wnt induced frizzled/Lrp complex formation. It was shown that LRP4 is an osteoblast expressed receptor for Dkk1 and sclerostin and regulates bone growth and bone turnover in vivo.3 In addition, it was shown that the extracellular domains of LRP4 and LRP1b proteins may function as a scavenger for signaling ligands or signal modulators in the extracellular space, thereby preserving signaling thresholds that are critical for embryonic development, as well as for the clear, but poorly understood role of LRPs in cancer.4
REFERENCES
1. Ohazama, A. et al: PLoS ONE 3:e4092, 2008
2. Kim, N. et al: Cell 135:335-42, 2008
3. Choi, H-Y. et al: PLoS ONE 4:e7930, 2009
4. Dietrich, M.F. et al: PLoS ONE 5:e9960, 2010
2. Kim, N. et al: Cell 135:335-42, 2008
3. Choi, H-Y. et al: PLoS ONE 4:e7930, 2009
4. Dietrich, M.F. et al: PLoS ONE 5:e9960, 2010
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CY1110 |
Antigen: | Human LRP4 C-terminal sequence |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
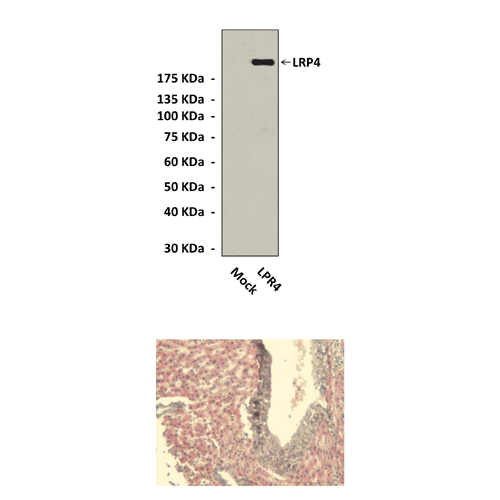
Predicted Molecular Weight of protein: | 220 kDa |
Specificity/Sensitivity: | Detects endogenous LRP4 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой