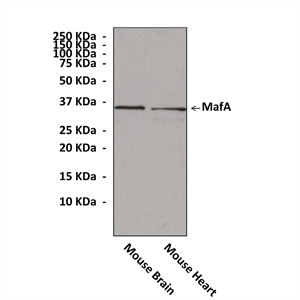
Anti-MafA: Rabbit MafA Antibody |
 |
BACKGROUND Maf proteins belong to a large class of transcription factors originally described as viral oncogenes. They are characterized by the presence of a basic leucine zipper (b-Zip) domain and the ability to bind to DNA MARE (Maf Recognition Elements) either as homodimers or heterodimers with other b-Zip proteins. These transcription factors have been associated with the regulation of multiple differentiation processes, including hematopoiesis, skin and lens development and hind-brain segmentation. The best characterized Maf factors expressed in the pancreas are MafA and MafB.1
MafA has been identified as an islet-enriched transcriptional activator that binds to the RIPE3b1 element in the promoter of the insulin gene and has been postulated to regulate insulin transcription in response to serum glucose level in β-cells of the pancreas. Unlike previously known islet-enriched transcriptional factors, such as Pdx1 and NeuroD/BETA2, which are expressed in non–β-cells as well as in β-cells, the expression of MafA is restricted to only β-cells, suggesting that MafA is responsible for tissue-specific expression of insulin. MafA is regulated by several intricate mechanisms. MafA undergoes extensive posttranslational modification by phosphorylation, ubiquitination and sumoylation, and these modifications regulate the turnover, DNA binding and transactivation function of the protein. Regulation of MafA expression is equally complex. The JNK/c-Jun pathway has been implicated in regulating MafA expression. The initial characterization of the β-cell-specific MafA promoter identified six conserved sequence domains. One of these regions in particular contains consensus motifs and binding sites for several β-cell-enriched transcription factors which ultimately play critical roles in controlling the expression of the gene. Interestingly, in cell culture, acute high glucose stimulation induces the accumulation of MafA, and MafA, in turn, regulates β-cell function. However, under chronic high glucose conditions, which occurs in the context of the diabetic state, β-cell function and, coincidentally, MafA levels decline. Currently, the mechanisms controlling the glucose-dependent accumulation of MafA are not well understood. It is suggested that MafA transcription is upregulated in β-cells acutely cultured in high glucose similar to what may occur in vivo under normoglycemic conditions.2 Under diabetic conditions, chronic hyperglycemia gradually deteriorates pancreatic beta-cell function, which is accompanied by decreased expression and/or DNA binding activities of MafA. Furthermore, MafA overexpression, together with PDX-1 and NeuroD, markedly induces insulin biosynthesis in various non-beta-cells and thereby is a useful tool to efficiently induce insulin-producing surrogate beta-cells. These results suggest that MafA plays a crucial role in pancreatic beta-cells and could be a novel therapeutic target for diabetes.3 In addition, it was reported that MafA is also expressed in the thymus and colocalizes with and modulates the expression of insulin in the thymus. Functional variants of mouse Mafa and human MAFA were identified and found to be associated with the expression level of insulin in the thymus and susceptibility to type 1 diabetes. MafA is therefore an important candidate among these transcriptional activators of insulin, leading to organ-specific autoimmunity to pancreatic β-cells by dysregulation of transcription of insulin in the thymus as an organ-specific self-antigen.4
REFERENCES
1. Aramata, S. et al: Endocrine J. 54:659-66, 2007
2. Vanderford, N.L.: Islets 3:35-7, 2011
3. Kaneto, H. et al: Adv. Drug Deliv. Rev. 61:489-96, 2009
4. Noso, S. et al: Diabetes 59:2579-87, 2010
2. Vanderford, N.L.: Islets 3:35-7, 2011
3. Kaneto, H. et al: Adv. Drug Deliv. Rev. 61:489-96, 2009
4. Noso, S. et al: Diabetes 59:2579-87, 2010
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CY1120 |
Antigen: | Raised against a short peptide from human MafA sequence. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 36 kDa |
Specificity/Sensitivity: | Detects endogenous MafA proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой