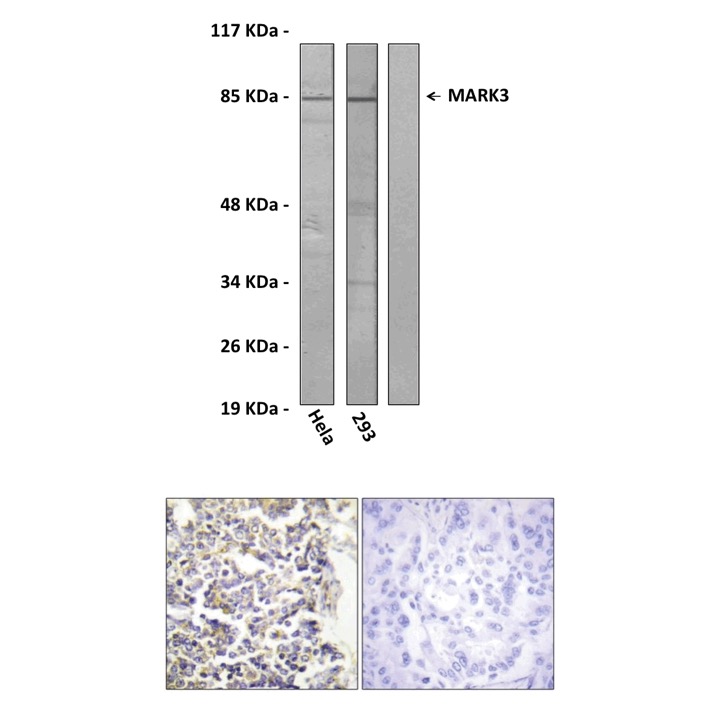
Anti-MARK3: Rabbit MARK3 Antibody |
 |
BACKGROUND Microtubules and their associated proteins (MAPs) play a key role in a dynamic network that involves the regulation of cell shape and cell polarity during differentiation, chromosome partitioning at mitosis, and intracellular transport. During these processes, the binding of tau and MAPs to microtubules is controlled by their phosphorylation state, which is meditated by microtubule-associated protein/microtubule affinity-regulating kinases (MARKs) (also known as partition-defective [Par]-1 kinase). MARKs are known to phosphorylate MAPs on their tubulin binding sites and consequently induce their detachment from microtubules Members of the MARK family were subsequently found to be highly conserved and to have key roles in cell processes such as determination of polarity, cell-cycle control, intracellular signal transduction, transport and cytoskeleton. This is important for neuronal differentiation, but is also prominent in neurodegenerative 'tauopathies' such as Alzheimer's disease. The identified functions of MARK/Par-1 are diverse and require accurate regulation.1,2 It was shown that MARK3 phosphorylates human Cdc25C on serine 216 and promotes 14-3-3 protein binding.3
There are four members of MARK/PAR-1 family in human: MARK1-4. The MARK/PAR-1 family proteins contain three conserved domains: an N-terminal catalytic domain, a central ubiquitin-associated (UBA) domain, and a C-terminal domain, which was termed the kinase-associated domain 1 (KA1). The KA1 domain was previously defined as comprising 50 amino acid residues by the Pfam database (accession no. PF02149), and all of the KA1 domains are localized at the C terminus and end in the highly conserved Glu-Leu-Lys-Leu motif, termed the ELKL motif. Several studies have suggested that this domain is involved in protein localization. On the other hand, the KA1 domain may be necessary for kinase autoinhibition. MARK contains several domains which offer multiple ways of regulation by posttranslational modification (e.g. phosphorylation), interactions with scaffolding proteins and subcellular targeting (e.g. 14-3-3), and interactions with other proteins, particularly the interactions between MARK and other kinases, such as MARKK/TAO-1 and PAK5. MARKK (a member of the Ste20 family of kinases) activates MARK by phosphorylating it at a critical threonine residue within the activation loop. Activated MARK in turn phosphorylates tau, causes its detachment from microtubules and renders them labile. PAK5 inactivates MARK, not by phosphorylation, but by binding to the catalytic domain. PAK5 contributes to microtubule stability by preventing the MARK-induced phosphorylation of tau; conversely, PAK5 contributes to actin dynamics, presumably through the activation of cofilin, an F-actin severing protein. Thus, MARK and its regulators MARKK and PAK5 appear to mediate the crosstalk between the actin and microtubule cytoskeleton in an antagonistic fashion.4
REFERENCES
1. Matenia, D. & Mandelkow. E.M. : Trends Biochem. Sci. 34:332-42, 2009
2. Drewes, G. et al: Trends Biochem. Sci. 23:307-11, 1998
3. Peng, C.Y. et al: Cell Growth Differ. 9:197-208, 1998
4. Timm, T. et al: Neurodegenerat. Dis. 3:207-17, 2006
2. Drewes, G. et al: Trends Biochem. Sci. 23:307-11, 1998
3. Peng, C.Y. et al: Cell Growth Differ. 9:197-208, 1998
4. Timm, T. et al: Neurodegenerat. Dis. 3:207-17, 2006
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CG1300 |
Antigen: | Synthesized peptide derived from human MARK3. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:500-1:1000 IP n/d IHC 1:50-1:100 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 86 kDa |
Specificity/Sensitivity: | Detects endogenous MARK3 proteins without cross-reactivity with other family memebers. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой