Anti-MBP-Tag: Mouse Maltose Binding Protein-Tag Antibody |
 |
BACKGROUND Maltose Binding Protein, MBP, is commonly used to create fusion proteins. The tag has the size of 392 amino acids (roughly 42 KDa),1 which, compared to other tags like the myc- or the FLAG-tag, is quite big. It is fused to the N-terminus of a protein. However, many commercially-available sources of MBP-tagged plasmids include a factor Xa site for cleavage of the MBP tag during protein purification.
A MBP-tag is often used to separate and purify as well as confirm expression of proteins that contain the MBP-fusion. MBP-fusion proteins can be produced in Escherichia coli, as recombinant proteins. The MBP part binds its substrate, amylose. Agarose beads can be coated with amylose, and such amylose-Agarose beads bind MBP-proteins. These beads are then washed, to remove contaminating bacterial proteins. Adding free amylose to beads that bind purified MBP-proteins will release the MBP-protein in solution. MBP-tag antibody is a useful tool for confirming protein expression, localization of expressed proteins in cells, as well as affinity-binding of MBP-tagged proteins.2
REFERENCES
1. Guan, C. et al. Gene 67: 21–30, 1987
2. Riggs, P., in Ausubel, F.M. et al. (eds), Cur¬rent Prot. in Molecular Biol. (1992) Greene Associates/Wiley Interscience, New York.
2. Riggs, P., in Ausubel, F.M. et al. (eds), Cur¬rent Prot. in Molecular Biol. (1992) Greene Associates/Wiley Interscience, New York.
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10165 |
Antigen: | Purified recombinant MBP-Tag expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | MBP-tag |
Applications & Suggested starting dilutions:* | WB 1:1000 IP 1:50 IHC n/d ICC 1:200 FACS 1:200 |
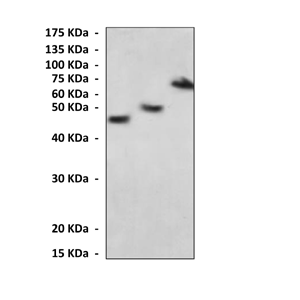
Predicted Molecular Weight of protein: | 42 kDa |
Specificity/Sensitivity: | Detects MBP-Tag proteins without cross-reactivity with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой