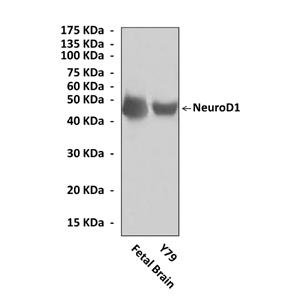
Anti-NeuroD1: Rabbit NeuroD1/Beta2 |
 |
BACKGROUND NeuroD1/beta2 is in the basic helix-loop-helix (bHLH) family of transcriptional activators and functions in a complex with the more generally distributed E2A (i.e. E47, E12, E2-5)-encoded proteins and HEB-encoded proteins. Dimerization between bHLH proteins depends on the HLH region, while protein-DNA interactions are mediated by the basic region. The presence of a tissue-enriched and a ubiquitously distributed bHLH factor in the activator complex is characteristic of other tissue-specific members of this family, the best characterized of which are involved in myogenic and neuronal differentiation. These activators bind to the consensus sequence CANNTG (termed an E-box), with heterodimerization increasing DNA binding and activation capacity.1
NeuroD1/beta2 is expressed in pancreatic islet endocrine cells, the intestine, the pituitary, and a subset of neurons in the central and peripheral nervous system. This factor was independently isolated and characterized by its ability to activate neurite formation upon ectopic expression in Xenopus embryos (termed NeuroD and referred to here as NeuroD1), and insulin reporter gene transcription in transfected beta cells (termed BETA2). NeuroD1/beta2 also stimulates secretin and proopiomelanocortin (POMC) transcription in the intestine and pituitary gland, respectively. Activation by NeuroD1/beta2 is potentiated by the p300/CREB binding protein (CBP) coactivator. Although the exact mechanism involved in p300/CBP-mediated transcription is unclear, it may result from bridging through direct interactions the activator to the basal transcriptional machinery and/or from promotion of a transcriptionally active state to targeted genes through its intrinsic histone acetyltransferase activity. Since p300/CBP also modulates the activity of a number of key activators involved in regulating cellular proliferation and differentiation, including the myogenic bHLH factors, its association with NeuroD1/BETA2 may be important for transcriptional signaling during development and in the adult.2
NeuroD1/beta2 is required during the development of specialized pancreatic and enteroendocrine cell types arising from gut endoderm, as well as being involved in differentiated gene product expression (i.e., insulin, secretin, and POMC). Furthermore, Neurod1 is essential for neuronal differentiation in the cerebellum and the ear and can convert non-neuronal cells into neurons through the regulation of over 500 downstream genes. It is known that neuronal and hair cell development of the inner ear critically depends on the basic Helix-Loop-Helix (bHLH) genes Neurog1 and Atoh1, respectively. Both genes regulated expression of NeuroD1 (Neurog1 in neurons and by Atoh1 in hair cells). Through this and other feedback, NeuroD1 suppresses hair cell differentiation in sensory ganglia and controls gene expression needed for outer hair cell maturation.3 Mutations in this gene result in type II diabetes mellitus and neurological abnormalities.4
REFERENCES
1. Liu, M. et al: Gene Dev. 14: 2839-54, 2000
2. Sharma, A. et al: Mol. Cell. Biol.19: 704-13, 1999
3. Janhan, I. et al: PloS ONE 5: 11661, 2010
4. Malecki, M.T. et al: Nature Genet. 23: 323-8, 1999
2. Sharma, A. et al: Mol. Cell. Biol.19: 704-13, 1999
3. Janhan, I. et al: PloS ONE 5: 11661, 2010
4. Malecki, M.T. et al: Nature Genet. 23: 323-8, 1999
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
|
Cat.No.:
|
CG1343
|
|
Antigen:
|
Short peptide from human NeuroD1/beta2 sequence.
|
|
Isotype:
|
Rabbit IgG
|
|
Species & predicted
species cross-
reactivity ( ):
|
Human, Mouse, Rat
|
|
Applications &
Suggested starting
dilutions:*
|
WB 1:1000 - 1:10000
IP 1:50
IHC n/d
ICC n/d
FACS n/d
|
|
Predicted Molecular
Weight of protein:
|
47 kDa
|
|
Specificity/Sensitivity:
|
Detects endogenous NeuroD1/beta2 proteins without cross-reactivity with other family members.
|
|
Storage:
|
Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles.
|
*Optimal working dilutions must be determined by end user.
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой