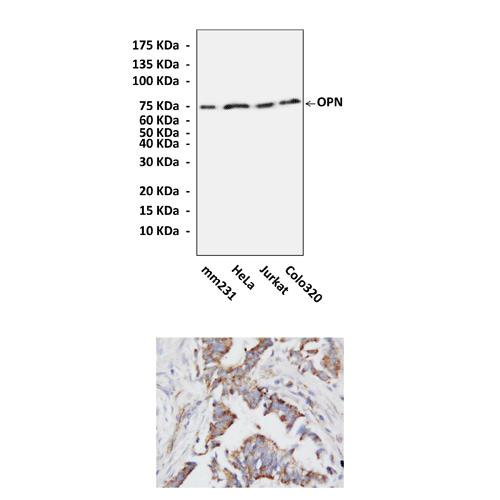
Anti-OPN: Rabbit OPN Antibody |
 |
BACKGROUND Osteopontin (OPN, also known as early T cell activation gene-1, or eta-1) is a secreted, multifunctional glycoprotein that exists either as a full-length molecule or as proteolytic fragments. Multiple forms of OPN have been described, including various glycosylated, phosphorylated, sulfated, and nonsulfated species, ranging from 25 to 75 kDa. OPN is synthesized by many cell types and involved in various physiologic and pathologic processes, including cell adhesion, migration, differentiation, apoptosis, angiogenesis, inflammatory responses, and tumor metastasis.1 The different effects that OPN elicit are attributable to its multiple receptors, binding sites, and its various forms. One of the major serine proteases to cleave Opn is thrombin, giving rise to a 24-kDa and a 45-kDa fragment. The 45-kDa fragment has multiple functional advantages in processes such as cell attachment, migration, and spreading through binding to alpha9beta1 and alpha4beta1. OPN is bound by multiple integrins, including alphavbeta3, alpha9beta1, alphavbeta5, alphavbeta1, alpha4beta1, alpha5beta1, and alpha4beta7. Specifically, alphavbeta3, alphavbeta5, alphavbeta6, and alphavbeta1 interact with the Arg-Gly-Asp (RGD) domain of OPN to influence cell adhesion and migration. In addition, there is evidence that CD44 can also bind OPN possibly via the v6 and v7 variants. Stem cells are known to express CD44 and α4 integrin, both of which are receptors capable of interacting with OPN. Indeed all these receptors mediate the adhesion of a broad range of cell types to OPN, which results in distinct cellular functions. For example, alphavbeta3 and alphavbeta1 mediate adhesion of smooth muscle cells, but only v 3 is involved in cell migration.2 OPN binds to cells, activating multiple and varied signaling pathways. It was reported that OPN upregulates alphavbeta3 integrin-mediated Janus kinase 2 (JAK2) phosphorylation and STAT3 activation in breast cancer.3
OPN was initially identified in osteoblasts as a mineralization-modulatory matrix protein. Now OPN has been studied as a multifunctional protein that is upregulated in a variety of acute and chronic inflammatory conditions, such as wound healing, fibrosis, autoimmune disease, and atherosclerosis. OPN is highly expressed at sites with atherosclerotic plaques, especially those associated with macrophages and foam cells. In the context of atherosclerosis, OPN is generally regarded as a proinflammatory and proatherogenic molecule. However, the role of OPN in vascular calcification (VC), which is closely related to chronic and active inflammation, is that of a negative regulator because it is an inhibitor of calcification and an active inducer of decalcification. In addition to being produced by cells of osteoblastic lineage, OPN has been shown to play important roles in chemotaxis, adhesion, and proliferation, all of which allow it to mediate inflammation and immunity to infectious diseases. For example, granulomatous responses are associated with high levels of OPN expression and OPN can function as a Th1 cell cytokine, enhancing IL-12 while inhibiting expression of the Th2 cell cytokine IL-10. Furthermore, OPN can alter the sensitivity of hematopoietic cells to other cytokine stimuli.4
OPN was initially identified in osteoblasts as a mineralization-modulatory matrix protein. Now OPN has been studied as a multifunctional protein that is upregulated in a variety of acute and chronic inflammatory conditions, such as wound healing, fibrosis, autoimmune disease, and atherosclerosis. OPN is highly expressed at sites with atherosclerotic plaques, especially those associated with macrophages and foam cells. In the context of atherosclerosis, OPN is generally regarded as a proinflammatory and proatherogenic molecule. However, the role of OPN in vascular calcification (VC), which is closely related to chronic and active inflammation, is that of a negative regulator because it is an inhibitor of calcification and an active inducer of decalcification. In addition to being produced by cells of osteoblastic lineage, OPN has been shown to play important roles in chemotaxis, adhesion, and proliferation, all of which allow it to mediate inflammation and immunity to infectious diseases. For example, granulomatous responses are associated with high levels of OPN expression and OPN can function as a Th1 cell cytokine, enhancing IL-12 while inhibiting expression of the Th2 cell cytokine IL-10. Furthermore, OPN can alter the sensitivity of hematopoietic cells to other cytokine stimuli.4
REFERENCES
1. Haylock, D.N. & Nilsson, S.K.: Br. J. Haematol. 134:467-74, 2006
2. Scatena, M. et al: Arterioscler Thromb Vasc Biol. 27:2302-9, 2007
3. Behera, R. et al: Carcinogen. 31:192-200, 2010
4. Cho, H.J. et al: Curr Atheroscler Rep. 11:206-13, 2009
2. Scatena, M. et al: Arterioscler Thromb Vasc Biol. 27:2302-9, 2007
3. Behera, R. et al: Carcinogen. 31:192-200, 2010
4. Cho, H.J. et al: Curr Atheroscler Rep. 11:206-13, 2009
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CA1431 |
Antigen: | A short peptide from human OPN carboxyl-terminal sequence. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Rabbit |
Applications & Suggested starting dilutions:* | WB 1:500 - 1:1000 IP n/d IHC 1:50 - 1:100 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 75 kDa |
Specificity/Sensitivity: | Detects endogenous OPN proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой