Anti-OTX2: Mouse OTX2 Antibody |
 |
BACKGROUND Orthodenticle homeobox 2 (OTX2; MIM 600037) belongs to the paired-class, bicoid subclass family of homeodomain genes, and is required for anterior brain and eye formation. Mammals have three OTX genes: OTX1, OTX2, and CRX. Mouse OTX1 and OTX2 are expressed in developing neural and sensory structures, including the brain, ear, nose, and eye. OTX2 expression is regulated in a spaciotemporal pattern with OTX2 expression proceeding and encompassing OTX1 expression domains during embryogenesis. OTX2 interacts with numerous proteins to coordinate cell determination and differentiation, including RX1, PAX6, SIX3, LHX2, MITF, GBX2, and HESX1. Homozygous OTX2 knockout embryos die in midgestation due to severe brain abnormalities, whereas heterozygotes display highly variable phenotypes, from normal to severe craniofacial malformations, depending on the genetic background. Conditional ablation of OTX2 in retinal neural cells leads to failure of photoreceptor and pineal gland development. In humans, loss-of-function mutations in OTX2 have been associated with anophthalmia and microphthalmia, with highly pleiotropic phenotypical expression within an affected family. Humans with heterozygous mutations in OTX2 were reported to have a variety of structural eye malformations but without craniofacial abnormalities.1
Otx2 is restricted to ventral tegmental area (VTA) neurons that are prevalently complementary to those expressing Girk2 and glycosylated active form of the dopamine transporter (Dat). It was shown that Otx2 is required to specify neuron subtype identity in VTA and may antagonize vulnerability to the Parkinsonian toxin MPTP.2 In addition, Otx2 is important for GnRH expression and direct interaction between Otx2 and Grg co-repressors regulates GnRH gene expression in hypothalamic neurons.3 Furthermore, one transcription factor that is expressed in retinal cell types (photoreceptors and retinal pigment epithelium), where abnormal function can lead to various diseases, is the homeoprotein Otx2. It is then necessary for neural retina development, maturation and photoreceptor fate determination, and for bipolar cell terminal differentiation after birth. It was shown that OTX2 is necessary for long-term maintenance of photoreceptors, likely through the control of specific activities of the RPE.4 Finally, Otx2 gene is amplified and overexpressed in medulloblastoma cell lines. Mechanistic investigations revealed upregulation of MYC as a potential mechanism whereby OTX2 promotes tumor progression. OTX2 is an important oncogenic driver in medulloblastoma.5 Other studies indicate that OTX2 directly induces a series of cell cycle genes but requires cooperating genes for an oncogenic acceleration of the cell cycle.
Otx2 is restricted to ventral tegmental area (VTA) neurons that are prevalently complementary to those expressing Girk2 and glycosylated active form of the dopamine transporter (Dat). It was shown that Otx2 is required to specify neuron subtype identity in VTA and may antagonize vulnerability to the Parkinsonian toxin MPTP.2 In addition, Otx2 is important for GnRH expression and direct interaction between Otx2 and Grg co-repressors regulates GnRH gene expression in hypothalamic neurons.3 Furthermore, one transcription factor that is expressed in retinal cell types (photoreceptors and retinal pigment epithelium), where abnormal function can lead to various diseases, is the homeoprotein Otx2. It is then necessary for neural retina development, maturation and photoreceptor fate determination, and for bipolar cell terminal differentiation after birth. It was shown that OTX2 is necessary for long-term maintenance of photoreceptors, likely through the control of specific activities of the RPE.4 Finally, Otx2 gene is amplified and overexpressed in medulloblastoma cell lines. Mechanistic investigations revealed upregulation of MYC as a potential mechanism whereby OTX2 promotes tumor progression. OTX2 is an important oncogenic driver in medulloblastoma.5 Other studies indicate that OTX2 directly induces a series of cell cycle genes but requires cooperating genes for an oncogenic acceleration of the cell cycle.
REFERENCES
1. Henderson, R.H. et al: Mol. Vis. 15:2442–7, 2009
2. Salvio, M.D. et al: Nature Neurosci. 13:1481-8, 2010
3. Larder, R. & Mellon, P.L.: J. Biol. Chem. 284:16966-78, 2008
4. Beby, F. et al: PLoS ONE 5(7): e11673, 2010
5. Adamson, D.C. et al: Cancer Res; 70: 181–9, 2010
2. Salvio, M.D. et al: Nature Neurosci. 13:1481-8, 2010
3. Larder, R. & Mellon, P.L.: J. Biol. Chem. 284:16966-78, 2008
4. Beby, F. et al: PLoS ONE 5(7): e11673, 2010
5. Adamson, D.C. et al: Cancer Res; 70: 181–9, 2010
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10390 |
Antigen: | Raised against recombinant human OTX2 fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:50 - 1:200 ICC 1:50 - 1:200 FACS 1:50 - 1:200 |
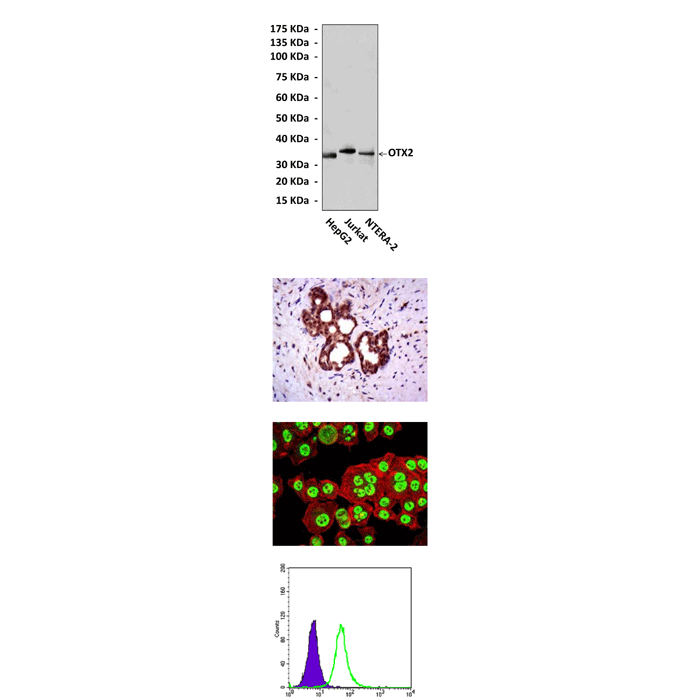
Predicted Molecular Weight of protein: | 32 kDa |
Specificity/Sensitivity: | Detects OTX2 proteins in various cell lysate. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой