Anti-OXR1: Rabbit Orexin Receptor 1 Antibody |
 |
BACKGROUND Orexins (also known as hypocretins) are lateral hypothalamic neuropeptides that are upregulated with fasting and can acutely promote appetite when administered into the central nervous system. Mammalian Orexin A is a 33-amino acid peptide with two intrachain disulphide bonds, whereas orexin B is a 28-amino acid, C-terminally amidated linear peptide. They both derive from the proteolytic cleavage of a common 130 amino acid precursor, the prepro-orexin. The orexins bind two G-coupled receptors, named orexin receptor 1 (ox1R) and orexin receptor 2 (ox2R), the first being highly selective for oxA and the second showing similar affinity for both peptides.1 The orexins are widely diffused in the neurons of the central nervous system, and in the intrinsic innervation of the gut. Recently, their presence has also been demonstrated in endocrine cytotypes belonging to the gastrointestinal tract, pancreas, adrenal glands, and testis. Thus, orexins are considered both neuropeptides and hormones. They are also normally found in the blood circulation of humans.The physiological activities of orexins in the mammalian body include the regulation of food intake, sleep–wake cycle, arterial blood pressure and heart rate, sexual behavior, and water assumption. The orexins have also been shown to influence the adrenal and gonadal functions acting on the respective neuroendocrine (NE) axes. Furthermore, convincing morpho-pharmacological evidence exists for their function as modulators of the action of neuropeptides and amines such as neuropeptide Y and 5-hydroxytryptamine (5-HT). The presence of oxA has been detected in the human placenta which is, however, a transient structure related only to pregnancy. Both orexin receptors are expressed in rat ovaries and fluctuate during the estrous cycle. The expression of prepro-orexin and of an orexin receptor sharing close similarities with ox2R has been described in the ovary of a non-mammalian species, the chicken. In addition, it was demonstrated the expression of OX1R and OX2R proteins were detected in the ovary of the pig and there was the impact of the hormonal milieu on the expression of both receptors. Thus, orexins may affect reproductive functions.2
Both OX1R and OX2R show differential affinity for the products of the prepro-orexin gene, and distinct expression patterns, indicating distinct roles in behavior and metabolism. The arcuate nucleus of hypothalamus (ARH) is a point of convergence for both orexin and leptin signaling, which modulate the activities of neuropeptide regulators of food intake and metabolism such as neuropeptide Y (NPY), agouti-related peptide (AGRP), and pro-opiomelanocortin (POMC). Pathologic leptin resistance may be mediated by changes in second messengers, including the long form of leptin receptor (LEPR), downstream signal transducer and activator of transcription-3 (STAT3), or the feedback suppressor of cytokine signal-3 (SOCS3). Central administration of orexin neuropeptides to rodents acutely promotes appetite, and prepro-orexin deficiency or postgestational ablation of orexin neurons in mice causes modest reductions in food intake. However, orexin-deficient mice also exhibit narcolepsy, inactivity, and obesity, indicating that orexin may exert an overall catabolic influence upon energy balance.3 Narcoleptic human individuals (the majority of which are orexin deficient) have also been reported to have greater body mass index and higher incidence of metabolic syndrome. This effect of orexin upon energy balance may be primary, since orexin-deficient narcoleptic patients showed higher body mass index than otherwise clinically indistinguishable narcoleptics with normal orexin levels. It is concluded that orexin promotes negative energy balance derives indirectly from loss-of-function studies. In addition, OX1R is equipped with a tyrosine-based inhibitory motif ITIM, which is tyrosine-phosphorylated on receptor activation, allowing the recruitment and activation of the tyrosine phosphatase SHP-2, leading to apoptosis.4
Both OX1R and OX2R show differential affinity for the products of the prepro-orexin gene, and distinct expression patterns, indicating distinct roles in behavior and metabolism. The arcuate nucleus of hypothalamus (ARH) is a point of convergence for both orexin and leptin signaling, which modulate the activities of neuropeptide regulators of food intake and metabolism such as neuropeptide Y (NPY), agouti-related peptide (AGRP), and pro-opiomelanocortin (POMC). Pathologic leptin resistance may be mediated by changes in second messengers, including the long form of leptin receptor (LEPR), downstream signal transducer and activator of transcription-3 (STAT3), or the feedback suppressor of cytokine signal-3 (SOCS3). Central administration of orexin neuropeptides to rodents acutely promotes appetite, and prepro-orexin deficiency or postgestational ablation of orexin neurons in mice causes modest reductions in food intake. However, orexin-deficient mice also exhibit narcolepsy, inactivity, and obesity, indicating that orexin may exert an overall catabolic influence upon energy balance.3 Narcoleptic human individuals (the majority of which are orexin deficient) have also been reported to have greater body mass index and higher incidence of metabolic syndrome. This effect of orexin upon energy balance may be primary, since orexin-deficient narcoleptic patients showed higher body mass index than otherwise clinically indistinguishable narcoleptics with normal orexin levels. It is concluded that orexin promotes negative energy balance derives indirectly from loss-of-function studies. In addition, OX1R is equipped with a tyrosine-based inhibitory motif ITIM, which is tyrosine-phosphorylated on receptor activation, allowing the recruitment and activation of the tyrosine phosphatase SHP-2, leading to apoptosis.4
REFERENCES
1. Voisin, T. et al: Cell Mol Life Sci. 60:72-87, 2003
2. Nitkiewicz, A. et al: Regulatory Peptides, 2010 (in press)
3. Korczynski, W.et al: J Physiol Pharmacol. 57 (Suppl 6):17-42, 2006
4. El Firar, A. et al: FASEB J. 23:4069-80, 2009
2. Nitkiewicz, A. et al: Regulatory Peptides, 2010 (in press)
3. Korczynski, W.et al: J Physiol Pharmacol. 57 (Suppl 6):17-42, 2006
4. El Firar, A. et al: FASEB J. 23:4069-80, 2009
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CA1100 |
Antigen: | Short peptide from human OXR1 sequence. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:50 - 1:200 ICC n/d FACS n/d |
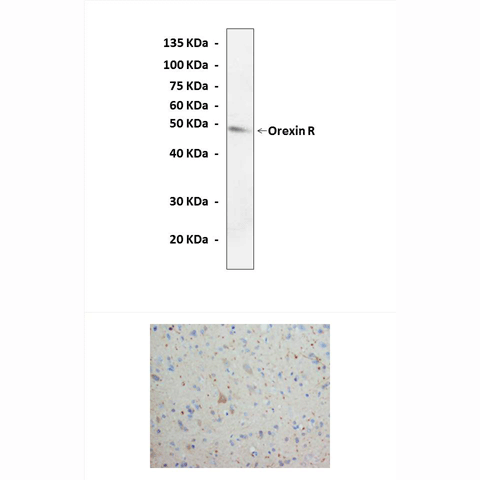
Predicted Molecular Weight of protein: | 48 kDa |
Specificity/Sensitivity: | Detects endogenous levels of OXR1 proteins without cross-reactivity with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой