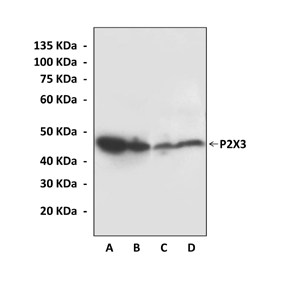
Anti-P2X3: Rabbit P2X3 Antibody |
 |
BACKGROUND Extracellular ATP is an important neurotransmitter signal in the brain as well as in vascular, immune, and endocrine cells. The purinergic system comprises receptors for extracellular ATP and adenosine, the P2 and P1 receptors, respectively. P2 purinergic receptors can be divided into metabotropic P2Y receptors (G protein coupled) and ionotropic P2X receptors (ligand-gated ion channels). The ionotropic P2X family comprises seven subtypes named P2X1–P2X7 that regulate cell function by opening cation channels permeable to Na+, K+, and Ca2+. Activation of these channels regulates the release of neurotransmitters and hormones, either through direct Ca2+ influx or by promoting membrane depolarization and thereby inducing action potentials.1
P2X receptors mediate a variety of physiological actions, including smooth muscle contraction, neuro-endocrine secretion and synaptic transmission. Among P2X receptors, P2X3 subunit-containing receptors (either homomeric or heteromeric combination of P2X3 and P2X2 receptors: P2X2/3) are expressed predominantly in nociceptive sensory neurons and are thought to mediate ATP nociceptive signaling. P2X3 receptor protein is exported from the soma of dorsal root ganglia (DRG) neurons to peripheral terminals in the skin and viscera and to central terminals projecting into inner lamina II of the dorsal horn of the spinal cord. ATP has been shown to depolarize isolated DRG neurons and excite the peripheral terminals of primary afferent neurons. In the spinal cord, ATP may act at presynaptic P2X3 receptors, facilitating glutamate release from primary afferents, as well as at postsynaptic receptors (including P2X1, P2X2, P2X4, P2X5, and P2X6 receptors) located on second-order neurons. Evidence for a direct physiological role of P2X3 homomeric and P2X2/3 heteromeric receptors has come from studies on P2X3 receptor null-mutant mice. These mice respond normally to acute noxious, thermal, and mechanical stimuli but display attenuated responses to non-noxious "warming" stimuli and show reduced formalin-induced nocifensive behaviors. In addition, their phenotype includes a marked urinary bladder hyporeflexia and, somewhat unexpectedly, increased hyperalgesia after peripheral inflammation of the hindpaw. However, ATP released from damaged or inflamed tissues can act at P2X receptors expressed on primary afferent neurones. The resulting depolarization can initiate action potentials that are interpreted centrally as pain. Indeed, P2X2/3 receptor was shown to be involved in the signaling of chronic inflammatory pain and some features of neuropathic pain.2 Recent evidence indicates that the strength of P2X3-mediated responses is modulated in vivo by altering the number of receptors at the plasma membrane. the level of functional receptor expressed on the cell surface is rapidly up-regulated in response to agonist stimulation, which also augments receptor endocytosis.3 It was also reported that Csk-mediated P2X3 receptor inhibition is a novel mechanism to limit over-activation of P2X3 receptors.1 Moreover, In humans, unlike rodents, P2X3 is thus not restricted to sensory neurones. Increased P2X3 in inflamed intestine suggests a potential role in dysmotility, for which it represents a new therapeutic target.4 Futhermore, it was reported that human beta cells express P2X3 and that activation of these receptors by ATP coreleased with insulin amplifies glucose-induced insulin secretion.5
P2X receptors mediate a variety of physiological actions, including smooth muscle contraction, neuro-endocrine secretion and synaptic transmission. Among P2X receptors, P2X3 subunit-containing receptors (either homomeric or heteromeric combination of P2X3 and P2X2 receptors: P2X2/3) are expressed predominantly in nociceptive sensory neurons and are thought to mediate ATP nociceptive signaling. P2X3 receptor protein is exported from the soma of dorsal root ganglia (DRG) neurons to peripheral terminals in the skin and viscera and to central terminals projecting into inner lamina II of the dorsal horn of the spinal cord. ATP has been shown to depolarize isolated DRG neurons and excite the peripheral terminals of primary afferent neurons. In the spinal cord, ATP may act at presynaptic P2X3 receptors, facilitating glutamate release from primary afferents, as well as at postsynaptic receptors (including P2X1, P2X2, P2X4, P2X5, and P2X6 receptors) located on second-order neurons. Evidence for a direct physiological role of P2X3 homomeric and P2X2/3 heteromeric receptors has come from studies on P2X3 receptor null-mutant mice. These mice respond normally to acute noxious, thermal, and mechanical stimuli but display attenuated responses to non-noxious "warming" stimuli and show reduced formalin-induced nocifensive behaviors. In addition, their phenotype includes a marked urinary bladder hyporeflexia and, somewhat unexpectedly, increased hyperalgesia after peripheral inflammation of the hindpaw. However, ATP released from damaged or inflamed tissues can act at P2X receptors expressed on primary afferent neurones. The resulting depolarization can initiate action potentials that are interpreted centrally as pain. Indeed, P2X2/3 receptor was shown to be involved in the signaling of chronic inflammatory pain and some features of neuropathic pain.2 Recent evidence indicates that the strength of P2X3-mediated responses is modulated in vivo by altering the number of receptors at the plasma membrane. the level of functional receptor expressed on the cell surface is rapidly up-regulated in response to agonist stimulation, which also augments receptor endocytosis.3 It was also reported that Csk-mediated P2X3 receptor inhibition is a novel mechanism to limit over-activation of P2X3 receptors.1 Moreover, In humans, unlike rodents, P2X3 is thus not restricted to sensory neurones. Increased P2X3 in inflamed intestine suggests a potential role in dysmotility, for which it represents a new therapeutic target.4 Futhermore, it was reported that human beta cells express P2X3 and that activation of these receptors by ATP coreleased with insulin amplifies glucose-induced insulin secretion.5
REFERENCES
1. D'Arco, M. et al: J. Biol. Chem. 284:21393-401, 2009
2. North, R.A. et al: J. Physiol. 554:301-8, 2004
3. Vacca, F. et al: J. Neurochem.109:1031-41, 2009
4. Yiangou, Y. et al: Neurogastroenterol. Motility 13: 365–369, 2001
5. Jacques-Silva, M.C. et al: Proc. Natl. Acad. Sci. USA 107:6465-70, 2010
2. North, R.A. et al: J. Physiol. 554:301-8, 2004
3. Vacca, F. et al: J. Neurochem.109:1031-41, 2009
4. Yiangou, Y. et al: Neurogastroenterol. Motility 13: 365–369, 2001
5. Jacques-Silva, M.C. et al: Proc. Natl. Acad. Sci. USA 107:6465-70, 2010
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CA1213 |
Antigen: | Short peptide from human P2X3 sequence. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:50 - 1:200 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 45 kDa |
Specificity/Sensitivity: | Detects endogenous levels of P2X3 proteins without cross-reactivity with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой