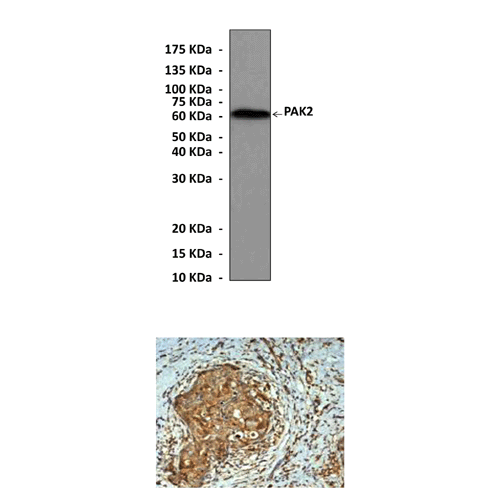
Anti-PAK2: Rabbit PAK2 Antibody |
 |
BACKGROUND The p21-activated kinases (Paks) are Ser/Thr protein kinases that bind to and, in some cases, are stimulated by activated forms of the small GTPases, Cdc42 and Rac. Paks are categorized into two subgroups based on architectural similarities. The conventional Group I Paks (Pak1, Pak2, Pak3) and nonconventional Group II Paks (Pak4, Pak5, Pak6). PAKs are stimulated dramatically by the GTP-bound forms of small GTPases, but also by a variety of GTPase-independent mechanisms. PAKs have an ancient origin and serve as important regulators of cytoskeletal dynamics and cell motility. In addition, they are implicated in transcription through MAPK cascades, death and survival signalling, and cell-cycle progression. Consequently, PAKs are implicated in a number of pathological conditions and in cell transformation. Paks function as downstream nodes for various oncogenic signaling pathways. Paks have also been shown to promote cell proliferation and accelerate mitotic abnormalities, which results in tumor formation and cell invasiveness.1
The conventional PAK family contains a conserved Cdc42/Rac-interacting binding domain (CRIB) that overlaps a kinase-inhibitory (KI) domain. There are three conserved proline-rich motifs that bind SH3 domain-containing proteins Nck, Grb2 and PIX. The group II PAKs (PAK4–PAK6) contains a CRIB sequence that binds GTPases, but lacks a KI domain. Upon Cdc42 (or related GTPase) binding, proteolysis or lipid binding, the kinase undergoes a conformational change that allows autophosphorylation. Phosphorylation of Ser-144 serves to disables the KI-domain–kinase interaction, while phosphorylation of Ser-198/203 reduces the affinity for PIX. Phosphorylation of the activation-loop Thr-422 may occur in trans, or may involve a third-party kinase such as PDK1. Other protein kinases might down-regulate PAK function: Akt phosphorylates PAK1 at Ser-21(Ser20 in PAK2), and this modification decreases binding of Nck to the PAK1 N-terminus while increasing kinase activity, which would suggest inhibition of PAK when Nck recruits PAK to various phosphotyrosine-containing complexes.2
RhoA, Rac1 and Cdc42 interact with a variety of associated kinases. Myosin light chain kinase (MLCK) is a calcium/calmodulin-responsive enzyme that maintains the myosin heavy chain–MLC complex in an active state, but is negatively regulated by PAK. Various studies have implicated ROK, MRCK and PAK in the regulation of LIMKs, which inactivate cofilin by phosphorylation at Ser-3. Once phosphorylated, cofilin/ADF (actin depolymerizing factor) can no longer bind effectively to F-actin, and the ability of these proteins to catalyze both F-actin depolymerization and severing is thus inhibited. PAK1 is thought to modulate R-MLC function primarily via inhibition of MLCK activity. Phosphorylation of MLCK occurs at Ser-439 and Ser-991; binding of calmodulin to MLCK is inhibited by modification of Ser-991. PAK1 has been shown to be able to bind to and regulate Ser-508 within the LIMK1 activation loop downstream of Rac1; Rho and Cdc42 are more closely linked to the effects of LIMK2. Thus the Rac and Cdc42 signaling pathways, acting via PAKs, can function either co-operatively with or antagonistically to Rho/ROK.3
The conventional PAK family contains a conserved Cdc42/Rac-interacting binding domain (CRIB) that overlaps a kinase-inhibitory (KI) domain. There are three conserved proline-rich motifs that bind SH3 domain-containing proteins Nck, Grb2 and PIX. The group II PAKs (PAK4–PAK6) contains a CRIB sequence that binds GTPases, but lacks a KI domain. Upon Cdc42 (or related GTPase) binding, proteolysis or lipid binding, the kinase undergoes a conformational change that allows autophosphorylation. Phosphorylation of Ser-144 serves to disables the KI-domain–kinase interaction, while phosphorylation of Ser-198/203 reduces the affinity for PIX. Phosphorylation of the activation-loop Thr-422 may occur in trans, or may involve a third-party kinase such as PDK1. Other protein kinases might down-regulate PAK function: Akt phosphorylates PAK1 at Ser-21(Ser20 in PAK2), and this modification decreases binding of Nck to the PAK1 N-terminus while increasing kinase activity, which would suggest inhibition of PAK when Nck recruits PAK to various phosphotyrosine-containing complexes.2
RhoA, Rac1 and Cdc42 interact with a variety of associated kinases. Myosin light chain kinase (MLCK) is a calcium/calmodulin-responsive enzyme that maintains the myosin heavy chain–MLC complex in an active state, but is negatively regulated by PAK. Various studies have implicated ROK, MRCK and PAK in the regulation of LIMKs, which inactivate cofilin by phosphorylation at Ser-3. Once phosphorylated, cofilin/ADF (actin depolymerizing factor) can no longer bind effectively to F-actin, and the ability of these proteins to catalyze both F-actin depolymerization and severing is thus inhibited. PAK1 is thought to modulate R-MLC function primarily via inhibition of MLCK activity. Phosphorylation of MLCK occurs at Ser-439 and Ser-991; binding of calmodulin to MLCK is inhibited by modification of Ser-991. PAK1 has been shown to be able to bind to and regulate Ser-508 within the LIMK1 activation loop downstream of Rac1; Rho and Cdc42 are more closely linked to the effects of LIMK2. Thus the Rac and Cdc42 signaling pathways, acting via PAKs, can function either co-operatively with or antagonistically to Rho/ROK.3
REFERENCES
1. Kumar, R. et al: Nature Rev. Cancer 6:459-71, 2006
2. Daniels, R. & Bokoch, G.M. et al: Trends Biochem. Sci. 24:350-5, 1999
3. Gerthoffer, W.T. & Gunst, S.J.: J. Appl. Physiol. 91:963-72, 2001
2. Daniels, R. & Bokoch, G.M. et al: Trends Biochem. Sci. 24:350-5, 1999
3. Gerthoffer, W.T. & Gunst, S.J.: J. Appl. Physiol. 91:963-72, 2001
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
|
Cat.No.:
|
CG1401
|
|
Antigen:
|
Raised against a short peptide from N-terminal sequence of human PAK2.
|
|
Isotype:
|
Rabbit IgG
|
|
Species & predicted
species cross-
reactivity ( ):
|
Human, Mouse, Rat
|
|
Applications &
Suggested starting
dilutions:*
|
WB 1:1000 - 1:10000
IP n/d
IHC 1:100 - 1:250
ICC 1:250 - 1:500
FACS 1:50
|
|
Predicted Molecular
Weight of protein:
|
62 kDa
|
|
Specificity/Sensitivity:
|
Detects endogenous PAK2 proteins without cross-reactivity with other family members.
|
|
Storage:
|
Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles.
|
*Optimal working dilutions must be determined by end user.
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой