Anti-RAG2: Mouse RAG2 Antibody |
 |
BACKGROUND During B and T cell development, the Ig and T cell receptor (TCR) variable-region genes are assembled from component germ line variable (V), diversity (D), and joining (J) gene segments through a process known as V(D)J recombination. Each V, D, and J gene segment is flanked by a recombination signal sequence (RSS) composed of conserved heptamer and nonamer sequences separated by either a 12- or 23-bp spacer. V(D)J recombination is initiated by the lymphoid-specific recombination-activating gene (RAG) 1 and 2 proteins, which recognize a pair of 12 and 23 RSSs and introduce a DNA double-strand break between each RSS and flanking coding sequence. Following cleavage by the RAG proteins, the liberated coding (and RS) ends are rejoined by a set of generally expressed proteins that carry out a nonhomologous end-joining reaction. The initiation of V(D)J recombination is tightly regulated via differential accessibility of antigen receptor loci and the lymphocyte-specific expression of RAG1 and RAG2. Both RAG1 and RAG2 are required for V(D)J recombination, because RAG1- or RAG2-deficient mice exhibit a complete block in lymphocyte development at the progenitor stage. In addition, RAG1 and RAG2 together are sufficient to initiate V(D)J recombination in vitro.1
RAG protein binding occurs in a highly focal manner to a small region of active chromatin encompassing Igκ and Tcrα J gene segments and Igh and Tcrβ J and J-proximal D gene segments. Formation of these small RAG-bound regions, referred to as recombination centers. Each RAG protein is independently capable of specific binding within recombination centers. While RAG1 binding was detected only at regions containing recombination signal sequences, RAG2 binds at thousands of sites in the genome containing histone 3 trimethylated at lysine 4.2 RAG2 contains a plant homeodomain (PHD) finger that specifically recognizes histone H3 trimethylated at lysine 4 (H3K4me3). Mutations that abrogate RAG2's recognition of H3K4me3 severely impair V(D)J recombination in vivo. Reducing the level of H3K4me3 similarly leads to a decrease in V(D)J recombination in vivo. RAG2 PHD finger couples histone H3 lysine 4 trimethylation with V(D)J recombination.3 Thus, the recombination centers coordinate V(D)J recombination by providing discrete sites within which gene segments are captured for recombination. Accumulation of the V(D)J recombinase protein RAG-2 is restricted to G0/G1 cells by phosphorylation-mediated degradation at the G1-S boundary. Cyclin A/CDK2 is shown to oppose RAG-2 accumulation; conversely, RAG-2 is induced by p27Kip1 and related CDK inhibitors. Coinduction of RAG-2 and G1 delay by p27Kip1 is accompanied by strong stimulation of V(D)J recombination. Unexpectedly, induction of RAG-2 accumulation in the absence of G1 delay has no effect on recombination frequency. p27Kip1 may stimulate V(D)J recombination by coordinating accumulation of RAG-2 with prolongation of G1, when nonhomologous end joining is preferentially active. Consistent with this, enforced expression of RAG-2 throughout cell cycle is associated with accumulation of aberrant recombination products reminiscent of those formed in the absence of nonhomologous end joining.4
RAG protein binding occurs in a highly focal manner to a small region of active chromatin encompassing Igκ and Tcrα J gene segments and Igh and Tcrβ J and J-proximal D gene segments. Formation of these small RAG-bound regions, referred to as recombination centers. Each RAG protein is independently capable of specific binding within recombination centers. While RAG1 binding was detected only at regions containing recombination signal sequences, RAG2 binds at thousands of sites in the genome containing histone 3 trimethylated at lysine 4.2 RAG2 contains a plant homeodomain (PHD) finger that specifically recognizes histone H3 trimethylated at lysine 4 (H3K4me3). Mutations that abrogate RAG2's recognition of H3K4me3 severely impair V(D)J recombination in vivo. Reducing the level of H3K4me3 similarly leads to a decrease in V(D)J recombination in vivo. RAG2 PHD finger couples histone H3 lysine 4 trimethylation with V(D)J recombination.3 Thus, the recombination centers coordinate V(D)J recombination by providing discrete sites within which gene segments are captured for recombination. Accumulation of the V(D)J recombinase protein RAG-2 is restricted to G0/G1 cells by phosphorylation-mediated degradation at the G1-S boundary. Cyclin A/CDK2 is shown to oppose RAG-2 accumulation; conversely, RAG-2 is induced by p27Kip1 and related CDK inhibitors. Coinduction of RAG-2 and G1 delay by p27Kip1 is accompanied by strong stimulation of V(D)J recombination. Unexpectedly, induction of RAG-2 accumulation in the absence of G1 delay has no effect on recombination frequency. p27Kip1 may stimulate V(D)J recombination by coordinating accumulation of RAG-2 with prolongation of G1, when nonhomologous end joining is preferentially active. Consistent with this, enforced expression of RAG-2 throughout cell cycle is associated with accumulation of aberrant recombination products reminiscent of those formed in the absence of nonhomologous end joining.4
REFERENCES
1. Akamatsu, Y. et al: Proc. Natl. Acad. Sci. USA 100:1209-14, 2003
2. Ji, Y. et al: Cell 141:419-31, 2010
3. Matthews, A.G.W. et al: Nature 450:1106-10, 2007
4. Lee, J. & Desiderio, S.: Immunity 11:771-81, 1999
2. Ji, Y. et al: Cell 141:419-31, 2010
3. Matthews, A.G.W. et al: Nature 450:1106-10, 2007
4. Lee, J. & Desiderio, S.: Immunity 11:771-81, 1999
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10343 |
Antigen: | Raised against recombinant human RAG2 fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
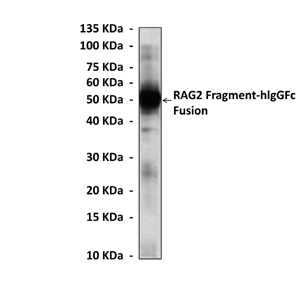
Predicted Molecular Weight of protein: | 59 kDa |
Specificity/Sensitivity: | Detects endogenous RAG2 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой