Anti-RANKL: Rabbit RANKL Antibody |
 |
BACKGROUND RANKL is a member of TNF superfamily. It is formed as a membrane-anchored molecule and can then be released from the cell after proteolytic cleavage by the metalloprotease desintegrin TNF-a convertase. RANKL is highly expressed on osteoblast/stromal cells, primitive mesenchymal cells surrounding the cartilaginous anlagen and hypertrophying chondrocytes. In addition to playing a role in the differentiation of osteoclasts from their precursor cells, RANKL also promotes increased activity and survival of these cells by an anti-apoptotic effect. The receptor activator of nuclear factor kappa B (RANK) is a member of the TNF receptor superfamily expressed on the surface of hematopoietic osteoclast progenitors, mature osteoclasts, chondrocytes, mammary gland epithelial cells, and trophoblast cells. It is a transmembrane heterotrimer containing three molecular intracellular domains (I, II and III). Binding of RANK with its cognate RANKL results in osteoclastogenesis by monocyte/macrophage progenitor differentiation to osteoclasts and the activation of mature osteoclasts. Activation of the RANK receptor on the osteoclast surface triggers intracellular signals mediated by the interaction of intracellular I, II and III domains and adapter proteins, TNF receptor-associated factors (TRAF). These TRAF-binding domains of the RANK molecule are important for the RANK-dependent induction of nuclear factor kappa B and JNK activities. RANKL also activates the anti-apoptotic serine/threonine kinase Akt/PKB through a signaling complex involving c-Src and TRAF6. c-Src and TRAF6 interact with each other and with RANK after receptor engagement and deficiency of c-Src or addition of Src family kinase inhibitors blocks RANK-mediated Akt/PKB activation in osteoclasts.1
In addition to its role in osteoclast development, RANKL was found to be expressed in activated T-cells, lymph nodes, spleen, thymus, intestinal lymphoid patches, and immature CD4/CD8 thymocytes . RANK is expressed on the surface of dendritic cells, mature T-cells and hematopoietic precursors, and its interaction with RANKL can induce Bcl-XL expression, CD40 expression and IL-12 production in dendritic cells; moreover, RANK/RANKL interaction can produce proliferation of T-cells activated by dendritic cells. In contrast to the CD40/CD40L system, RANK/RANKL signaling does not affect the expression of surface molecules, and maximum levels of RANKL are attained 48 h after the initial activation of T-cells (and sustained for 96 h), while CD40L is rapidly expressed and down-regulated . This suggests that CD40/CD40L interactions might control the initial phases of the response, while RANK/RANKL might act at later time points. RANK and RANKL are also important factors in lymphoid tissue development and in maturation of T- and B-cell precursors in bone marrow. In addition, Osteoprotegerin (OPG) is a protein with homology to members of the TNF receptor family and is produced and released by activated osteoblast cells. OPG functions as a soluble decoy receptor for RANKL and competes with RANK for RANKL binding. Consequently, OPG is an effective inhibitor of osteoclast maturation and osteoclast activation in vitro and in vivo.2 Moreover it was shown that RANKL can induced by or stimulate immune competent cells to produce various immune cytokines. Thus, given the important role of RANK, RANKL and OPG in bone metabolism and immune function, it has been suggested that the RANK/RANKL/OPG system and immune cytokines including TNF-a, IL-1, IL-11, IL-17, and others may work together in order to cause bone resorption via regulation of the RANKL/OPG ratio.3
In addition to its role in osteoclast development, RANKL was found to be expressed in activated T-cells, lymph nodes, spleen, thymus, intestinal lymphoid patches, and immature CD4/CD8 thymocytes . RANK is expressed on the surface of dendritic cells, mature T-cells and hematopoietic precursors, and its interaction with RANKL can induce Bcl-XL expression, CD40 expression and IL-12 production in dendritic cells; moreover, RANK/RANKL interaction can produce proliferation of T-cells activated by dendritic cells. In contrast to the CD40/CD40L system, RANK/RANKL signaling does not affect the expression of surface molecules, and maximum levels of RANKL are attained 48 h after the initial activation of T-cells (and sustained for 96 h), while CD40L is rapidly expressed and down-regulated . This suggests that CD40/CD40L interactions might control the initial phases of the response, while RANK/RANKL might act at later time points. RANK and RANKL are also important factors in lymphoid tissue development and in maturation of T- and B-cell precursors in bone marrow. In addition, Osteoprotegerin (OPG) is a protein with homology to members of the TNF receptor family and is produced and released by activated osteoblast cells. OPG functions as a soluble decoy receptor for RANKL and competes with RANK for RANKL binding. Consequently, OPG is an effective inhibitor of osteoclast maturation and osteoclast activation in vitro and in vivo.2 Moreover it was shown that RANKL can induced by or stimulate immune competent cells to produce various immune cytokines. Thus, given the important role of RANK, RANKL and OPG in bone metabolism and immune function, it has been suggested that the RANK/RANKL/OPG system and immune cytokines including TNF-a, IL-1, IL-11, IL-17, and others may work together in order to cause bone resorption via regulation of the RANKL/OPG ratio.3
REFERENCES
1. Boyce, B.F. & Xing, L.: Arthritis Res Ther. 9 (Suppl 1):S1, 2007
2. Baud’huin, M et al:Cell. Mol. Life Sci. 64:2334-50, 2007
3. Tanaka, S. et al: Immunol. Rev. 208:30-49, 2005
2. Baud’huin, M et al:Cell. Mol. Life Sci. 64:2334-50, 2007
3. Tanaka, S. et al: Immunol. Rev. 208:30-49, 2005
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
|
Cat.No.:
|
CG1726
|
|
Antigen:
|
Short peptide from human RANKL sequence.
|
|
Isotype:
|
Rabbit IgG
|
|
Species & predicted
species cross-
reactivity ( ):
|
Human
|
|
Applications &
Suggested starting
dilutions:*
|
WB 1:500 - 1:3000
IP n/d
IHC n/d
ICC n/d
FACS n/d
|
|
Predicted Molecular
Weight of protein:
|
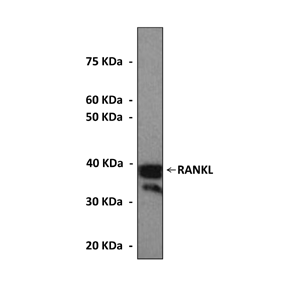
38 kDa
|
|
Specificity/Sensitivity:
|
Detects endogenous human RANKL proteins without cross-reactivity with other family.
|
|
Storage:
|
Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles.
|
*Optimal working dilutions must be determined by end user.
Extended Family Products
|
| ||||||||||
| ||||||||||
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой