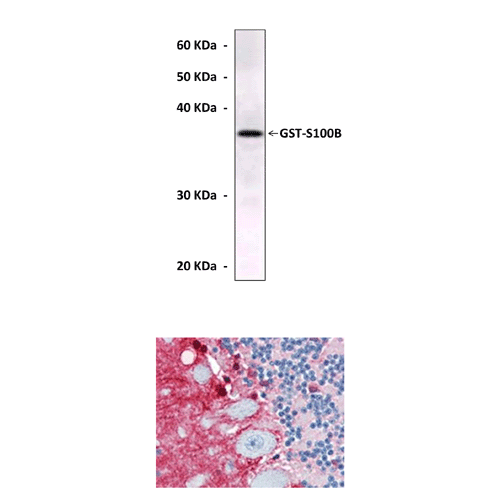
Anti-S100B: Mouse S100B Antibody |
 |
BACKGROUND S100 proteins are small, often acidic polypeptides of approximately 10 kDa containing an N-terminal and a C-terminal EF hand separated by a rather unstructured linker region. They were first identified in the brain and named S100 because of their solubility in 100% saturated ammonium sulfate. In humans, 19 family members are currently known with most S100 genes clustered on human chromosome 1q21 (S100A1 to S100A16). S100 proteins encoded elsewhere in the genome are S100B, S100P and S100Z. S100 proteins typically form homo- and, in some cases, also heterodimers. The dimers are thought to represent the functionally relevant entity because dimer formation appears to be required for effector protein binding. Upon binding of Ca2+, relatively large conformational changes occur, and hydrophobic surfaces are exposed in the dimer, allowing the interaction with a variety of cellular target (effector) proteins. A variety of such interaction partners have been identified, albeit in most cases still without proof of a physiological significance of the respective interaction observed in biochemical approaches. Thus, S100 proteins are considered to be signal transmitters that respond to intracellular Ca2+ rises by binding to cellular target proteins and modulating their activity.1
S100B is a member of the S100 family of proteins. The highest level of expression of the S100B protein is in the brain and is found primarily in the cytoplasm of astrocytes. Results of in vitro studies suggest a variety of intracellular functions of S100B, including cell growth, cell structure, energy metabolism, and calcium homeostasis. S100B is secreted from astrocytes, suggesting that it might also have extracellular functions. Exogenous S100B increases intracellular calcium concentrations in both cultured neurons and astrocytes. Elevated neuronal calcium might affect calcium-dependent processes involved in synaptic plasticity.2 In addition S100B is involved in regulation of cell cycle progression and cell differentiation. A Ca2+ dependent conformational change in dimeric S100B (beta-beta) is required for it to bind p53 and inhibit phosphorylation of this tumor suppressor in its C-terminal negative regulatory domain. Moreover, S100B and p53 interaction not only induces total inhibition of p53 oligomerization but also promotes disassembly of the p53 oligomers.3
S100B is a member of the S100 family of proteins. The highest level of expression of the S100B protein is in the brain and is found primarily in the cytoplasm of astrocytes. Results of in vitro studies suggest a variety of intracellular functions of S100B, including cell growth, cell structure, energy metabolism, and calcium homeostasis. S100B is secreted from astrocytes, suggesting that it might also have extracellular functions. Exogenous S100B increases intracellular calcium concentrations in both cultured neurons and astrocytes. Elevated neuronal calcium might affect calcium-dependent processes involved in synaptic plasticity.2 In addition S100B is involved in regulation of cell cycle progression and cell differentiation. A Ca2+ dependent conformational change in dimeric S100B (beta-beta) is required for it to bind p53 and inhibit phosphorylation of this tumor suppressor in its C-terminal negative regulatory domain. Moreover, S100B and p53 interaction not only induces total inhibition of p53 oligomerization but also promotes disassembly of the p53 oligomers.3
REFERENCES
1. Schäfer, B.W. & Heizmann, C.W.:Trends Biochem. Sci. 21:134-40, 1996
2. Nishiyama, H. et al: Proc. Natl. Acad. Sci. USA 99:4037-42, 2002
3. Rust, R.R. et al: Nature Struct. Biol. 7:570-4, 2000
2. Nishiyama, H. et al: Proc. Natl. Acad. Sci. USA 99:4037-42, 2002
3. Rust, R.R. et al: Nature Struct. Biol. 7:570-4, 2000
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10223 |
Antigen: | Purified recombinant human S100B proteins expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:200 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 11 kDa |
Specificity/Sensitivity: | Detects S100B proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой