Anti-uPAR: Rabbit uPAR/PLAUR Antibody |
 |
BACKGROUND The urokinase plasminogen activator receptor (uPAR, or PLAUR) has been implicated in the growth, metastasis, and angiogenesis of several solid and hemotologic malignancies. uPAR/PLAUR is part of a cell surface system that also consists of the serine protease uPA and several specific inhibitors (plasminogen activator inhibitors 1 and 2).1 This system has classically been thought to drive tumor progression by mediating directed extracellular proteolysis on the surface of migrating or invading cells. uPA can bind to uPAR/PLAUR leading to the subsequent activation of plasminogen to plasmin. Plasmin is a promiscuous protease that initiates several extracellular proteolytic cascades. The binding of uPA to uPAR/PLAUR increases the efficiency of plasminogen activation and also serves to localize these proteolytic cascades to the migrating or invading edge of cells. This proteolysis is tightly controlled by PAI-1 and PAI-2, and the PAI-uPA- uPAR/PLAUR complex can be internalized with uPAR/PLAUR being recycled to the cell surface.2 Thus, uPA, uPAR/PLAUR, and PAI-1 can all potentially be targeted for cancer therapy. In addition to mediating proteolysis, this receptor appears to also mediate cell signaling, proliferation, and survival.
REFERENCES
1. Blasi, F. & Carmeliet, P.: Nat. Rev. Mol. Cell. Biol. 3:932-43, 2002
2. Mazar, A.P. : Clin Cancer Res. 14:5649-55, 2008
2. Mazar, A.P. : Clin Cancer Res. 14:5649-55, 2008
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CA1344 |
Antigen: | Short peptide from human uPAR/PLAUR sequence. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
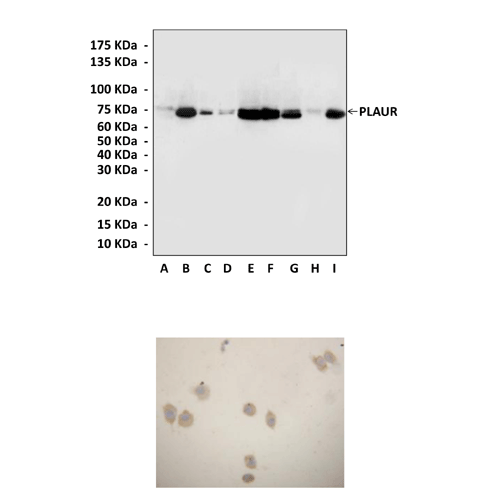
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:50 - 1:200 ICC 1:50 - 1:200 FACS n/d |
Predicted Molecular Weight of protein: | 70 kDa |
Specificity/Sensitivity: | Detects endogenous levels of uPAR/PLAUR proteins without cross-reactivity with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой