Anti-VEGFR1: Polyclonal VEGF Receptor 1 Antibody |
 |
BACKGROUND The biological functions of VEGF polypeptides are mediated upon binding to type III receptor tyrosine kinases (RTKs), VEGFR-1 (Flt-1), VEGFR-2 (KDR/Flk-1) and VEGFR-3 (Flt-4). VEGF receptors are closely related to Fms, Kit and PDGF receptors. They consist of seven extracellular immunoglobulin (Ig)-like domains, a transmembrane (TM) domain, a regulatory juxtamembrane domain, an intracellular tyrosine kinase domain interrupted by a short peptide, the kinase insert domain, followed by a sequence carrying several tyrosine residues involved in recruiting downstream signaling molecules.The binding of VEGF to the receptors induces dimerization and transphosphorylation of the receptors, followed by tyrosine phosphorylation and/or activation of several downstream substrates including the second messenger-producing enzymes phospholipase C-gamma and phosphotidylinositol 3-kinase, p120GAP, the adaptor proteins Nck, Grb2 and Shc, the tyrosine phosphatases SHP1 and SHP2 and the cytoskeleton associated proteins FAK and paxillin, these events leading to the activation of the extracellular-signal regulated kinase and p38 stress-activated protein kinase pathway.1 Although both VEGFR1 and VEGFR2 are expressed in the vascular endothelium, the angiogenic signals of VEGFs are mainly transduced by VEGFR2, which stimulate the proliferation, survival, migration, and permeability of endothelial cells. VEGFR1 has much higher binding affinity to VEGF ligands, but much lower tyrosine kinase activitiy than VEGFR2.2 There are evidences that suggested soluble VEGFR1 variant (sFlt1) acts as a decoy by binding and neutralizing VEGF in the absence of catalytic activation.3 More recent data indicate that the kinase activity of VEGFR-1 does play an essential role during pathological angiogenesis and in wound healing, by potentiating VEGFR-2 signaling, however, the molecular details for this receptor cross-talk have not yet been elucidated. In addition to endothelial cells, VEGFR-1 is expressed in bone marrow (BM) progenitor cells, myelomonocytic inflammatory cells, and vascular smooth muscle cells (SMCs). VEGFR-1 in myeloid cells mediates monocyte migration and has a regulatory role in the development of inflammatory diseases such as rheumatoid arthritis and arteriosclerosis.2
REFERENCES
1. Olsson AK et al.: Nature Rev Cell Biol. 7:359-371, 2006.
2. Cébe-Suarez S et al.: Cell. Mol. Life Sci. 63:601–615, 2006.
3. Shibuya M: Angiogenesis 9:225-30, 2006.
2. Cébe-Suarez S et al.: Cell. Mol. Life Sci. 63:601–615, 2006.
3. Shibuya M: Angiogenesis 9:225-30, 2006.
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
|
Cat.No.:
|
CB3839
|
|
Antigen:
|
Sequence near the human VEGFR1 carboxyl terminal.
|
|
Isotype:
|
Affinity Purified Rabbit IgG
|
|
Species & predicted
species cross-
reactivity ( ):
|
Human, Mouse
|
|
Applications &
Suggested starting
dilutions:
|
WB 1:1000
IP n/d IHC (Paraffin) n/d ICC n/d
FACS n/d
|
|
Predicted Molecular
Weight of protein:
|
180 kDa
|
|
Specificity/Sensitivity:
|
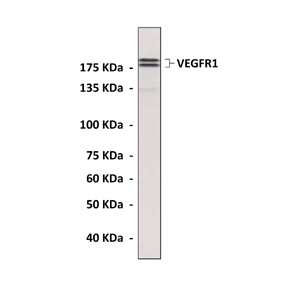
Detects endogenous levels of VEGFR1 proteins in endothelial cell lysates.
|
|
Storage:
|
Store at 4° C for frequent use; at -20° C for at least one year.
|
*Optimal working dilutions must be determined by end user.
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой