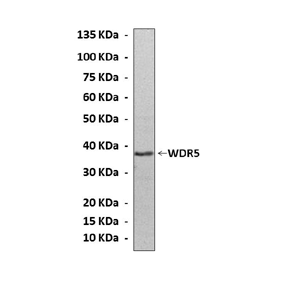
Anti-WDR5: Mouse WDR5 Anitbody |
 |
BACKGROUND The WD40-repeat proteins are a large family of proteins found in all eukaryotes and implicated in very diverse functions such as signal transduction, vesicular trafficking, cytoskeletal assembly, cell cycle control, apoptosis and transcription regulation. The common function of these WD40-repeat proteins is to serve as a rigid scaffold for protein–protein interaction, and to coordinate downstream events, such as ubiquitination. WDR5, a BMP-2-induced gene, is a WD repeat protein that is essential for histone H3 lysine 4 (H3K4) trimethylation, a marker of actively transcribed genes. It was shown that WDR5 is a core subunit of the human MLL and SET1 (hCOMPASS) histone H3 Lys4 (H3K4) methyltransferase complexes and directly associates with K4-dimethylated H3 in vitro and in vivo. WDR5 is required for recruiting the methyltransferase complexes to the K4-dimethylated H3 tail and is involved in di- to trimethylation conversion of histone H3 at K4. H3K4me2 appeared to interact with WDR5 via nonconventional hydrogen bonds between the two -methyl groups of the dimethylated K4 and the carboxylate oxygen of Glu322 in WDR5. Although initial studies suggested that WDR5 interacts with methylated H3K4 to catalyze Lys4 trimethylation, recent work has revealed that it binds an arginine-bearing motif in MLL1, promoting complex assembly and activity. These findings suggest that WDR5 functions as a peptidyl arginine–recognition factor that facilitates the assembly of hCOMPASS and other chromatin-modifying complexes.1 In addition to its role in histone methylation, WDR5 and WDS, the Drosophila homolog of WDR5, are associated with several histone acetyltransferase (HAT) complexes, including the MSL (male-specific lethal) complex; the ATAC (Ada Two A-containing) complex, which functions as both an H3 and H4 HAT; and MOF, a H4K16 acetyltransferase complex. Thus, WDR5 plays important roles in multiple complexes that display distinct catalytic activities on histone substrates.2
WDR5 accelerates osteoblast and chondrocyte differentiation in vitro, and is developmentally expressed in osteoblasts as well as in proliferating and hypertrophic chondrocytes. Targeted expression of WDR5 to osteoblasts resulted in earlier activation of the canonical Wnt signaling pathway in the bone collar as well as in primary calvarial osteoblast cultures. In addition, overexpression of WDR5 increased the expression of OPG, a target of the canonical Wnt signaling pathway. Thus, WDR5 accelerates osteoblast differentiation in association with activation of the canonical Wnt pathway.3 It was also shown that during chicken limb development, WDR5 is required for endochondral bone formation and that WDR5 influences this process, at least in part, by regulating Runx2 expression.4
WDR5 accelerates osteoblast and chondrocyte differentiation in vitro, and is developmentally expressed in osteoblasts as well as in proliferating and hypertrophic chondrocytes. Targeted expression of WDR5 to osteoblasts resulted in earlier activation of the canonical Wnt signaling pathway in the bone collar as well as in primary calvarial osteoblast cultures. In addition, overexpression of WDR5 increased the expression of OPG, a target of the canonical Wnt signaling pathway. Thus, WDR5 accelerates osteoblast differentiation in association with activation of the canonical Wnt pathway.3 It was also shown that during chicken limb development, WDR5 is required for endochondral bone formation and that WDR5 influences this process, at least in part, by regulating Runx2 expression.4
REFERENCES
1. Trievel, R.C. & Shilatifard, A.: Nat. Struct. Mol. Biol. 16: 678-80, 2009
2. Suganuma, T. et al; Gene Dev. 22:1265-8, 2008
3. Gori, F. et al: Dev. Biol. 295:498-506, 2006
4. Zhu, S. et al: J. Bone Mineral Res. 25:240414, 2010
2. Suganuma, T. et al; Gene Dev. 22:1265-8, 2008
3. Gori, F. et al: Dev. Biol. 295:498-506, 2006
4. Zhu, S. et al: J. Bone Mineral Res. 25:240414, 2010
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10264 |
Antigen: | Purified recombinant human WDR5 fragments expressed in E. coli. |
Isotype: | Mouse IgG2b |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP 1:50 IHC n/d ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 37 kDa |
Specificity/Sensitivity: | Detects WDR5 proteins without cross-reactivity with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой