EG-VEGF ELISA Kit, Human |
 |
BACKGROUND Endocrine gland-derived vascular endothelial growth factor (EG-VEGF) is a 105-amino acid protein (including signal sequence) with 10 cysteines and is highly related to a protein (venom protein A) found in abundance in the venom of black mamba snake (Dendroaspis polylepsis). It belongs to the AVIT protein family and is identical with prokineticin-1(PK1). It has been demonstrated that EG-VEGF/PK1 is an endothelial cell mitogen with a unique selectivity, essentially limited to endothelial cells derived from endocrine organs. Expression of human EG-VEGF is restricted to the steroidogenic glands, ovary, testis, adrenal and placenta and is often complementary to the expression of VEGF. Moreover, it was shown that the mouse ortholog of EG-VEGF has 88 % protein sequence similarity with human EG-VEGF. Mouse EG-VEGF are expressed predominantly in hepatocytes and renal tubule cells. The mouse EG-VEGF promoter appears to lack the binding site for a transcription factor involved in steroidogenic-specific transcription. Within liver and kidney, EG-VEGF receptor expression is restricted largely to endothelial cells. Mouse EG-VEGF promotes proliferation and survival of endothelial cells.1
It was demonstrated that EG-VEGF is a potent survival factor for bovine adrenal cortex-derived endothelial cells. The expression of EG-VEGF was found in the adrenal cortex. Glomerulosa and fasciculata cells express EG-VEGF and its receptors. EG-VEGF has no effect on the steroidogenic activity of bovine glomerulosa cells and fasciculata cells but appears to be mitogenic for both cell types and thus acts as an autocrine mitogen for endocrine cells. EG-VEGF resembles VEGF in that it causes extensive angiogenesis and cyst formation when delivered in the ovary. EG-VEGF differs from VEGF in that it does not promote angiogenesis in the cornea or skeletal muscle. Two receptors have been characterized and termed PK2 (PKR1 and PKR2, respectively).2 PKR1 and PKR2 are G protein-coupled receptors (GPCR), which also bind the related factor prokineticin-2(PK2). The receptors bind and respond to the EG-VEGF/PK1 and PK2 by mobilization of calcium, turnover of phosphoinositide, and activation of mitogen-activated protein kinase (MAPK) signaling pathways. Both receptors were shown to be expressed in endocrine and nonendocrine tissues by reverse transcriptase-polymerase chain reaction (PCR) of cDNA. It was recently shown that PK2 is expressed highly at inflammatory sites and can stimulate monocyte migration and survival. It was also shown that EG-VEGF binds to murine macrophages and induces differentiation of human and mouse bone marrow-derived cells into the monocyte/macrophage lineage.3 In addition, EG-VEGF has also been shown to directly induce contractility of smooth muscles. Moreover, EG-VEGF plays important role in regulation of Dickkopf 1 (DKK1) expression, a negative regulator of canonical Wnt signaling, and its function in the non-pregnant endometrium and first trimester decidua.4
It was demonstrated that EG-VEGF is a potent survival factor for bovine adrenal cortex-derived endothelial cells. The expression of EG-VEGF was found in the adrenal cortex. Glomerulosa and fasciculata cells express EG-VEGF and its receptors. EG-VEGF has no effect on the steroidogenic activity of bovine glomerulosa cells and fasciculata cells but appears to be mitogenic for both cell types and thus acts as an autocrine mitogen for endocrine cells. EG-VEGF resembles VEGF in that it causes extensive angiogenesis and cyst formation when delivered in the ovary. EG-VEGF differs from VEGF in that it does not promote angiogenesis in the cornea or skeletal muscle. Two receptors have been characterized and termed PK2 (PKR1 and PKR2, respectively).2 PKR1 and PKR2 are G protein-coupled receptors (GPCR), which also bind the related factor prokineticin-2(PK2). The receptors bind and respond to the EG-VEGF/PK1 and PK2 by mobilization of calcium, turnover of phosphoinositide, and activation of mitogen-activated protein kinase (MAPK) signaling pathways. Both receptors were shown to be expressed in endocrine and nonendocrine tissues by reverse transcriptase-polymerase chain reaction (PCR) of cDNA. It was recently shown that PK2 is expressed highly at inflammatory sites and can stimulate monocyte migration and survival. It was also shown that EG-VEGF binds to murine macrophages and induces differentiation of human and mouse bone marrow-derived cells into the monocyte/macrophage lineage.3 In addition, EG-VEGF has also been shown to directly induce contractility of smooth muscles. Moreover, EG-VEGF plays important role in regulation of Dickkopf 1 (DKK1) expression, a negative regulator of canonical Wnt signaling, and its function in the non-pregnant endometrium and first trimester decidua.4
REFERENCES
1. Brouillet, S. et al: Mol. Biol. Cell 21:2832-43, 2010
2. Negri, N. et al: Int. Rev. Neurobiol. 85:145-57, 2009
3. Dorsch, M. et al: J. Leuk. Biol. 78:426-34, 2005
4. Macdonald, L.J. et al: Mol. Hum. Reprod. 2011 (In Press)
2. Negri, N. et al: Int. Rev. Neurobiol. 85:145-57, 2009
3. Dorsch, M. et al: J. Leuk. Biol. 78:426-34, 2005
4. Macdonald, L.J. et al: Mol. Hum. Reprod. 2011 (In Press)
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CL0575 |
Target Protein Species: | Human |
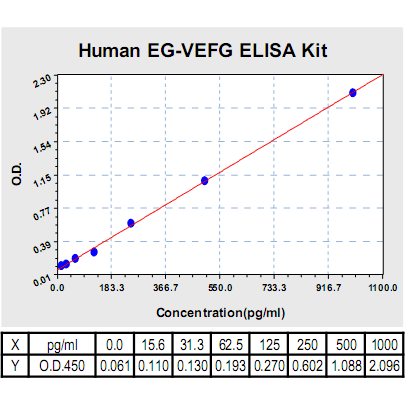
Range: | 15.6pg/ml-1000pg/ml |
Specificity: | No detectable cross-reactivity with any other cytokine. |
Storage: | Store at 4°C. Use within 6 months. |
ELISA Kits are based on standard sandwich enzyme-linked immunosorbent assay technology. Freshly prepared standards, samples, and solutions are recommended for best results.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой