Lipocalin-2/NGAL ELISA Kit, Mouse |
 |
BACKGROUND Lipocalins comprise a family of diverse proteins that exhibit only limited amino acid sequence similarity but share a common tertiary structure. Lipocalins are typically small (160–180 aa) secreted proteins that have several common molecular properties. Lipocalins bind with high affinity to lipophilic molecules (such as retinoids, fatty acids, cholesterols, and prostaglandins); and they form covalent and noncovalent complexes with other soluble macromolecules (e.g., neutrophil gelatinase). Initially characterized as transporter proteins, it is now well established that the Lipocalins perform a variety of important functions. For instance, several Lipocalins (including apolipoprotein D, quiescence-specific protein, purpurin, and alpha1-microglobulin) have been implicated in the modulation of cell growth and metabolism, whereas others (alpha1-microglobulin, glycodelin, and Lipocalin 2) appear to regulate the immune response.1
Lipocalin 2 (Lcn2)—also known as neutrophil gelatinase–associated Lipocalin, siderocalin, and 24p3. It was originally described as an acute-phase protein in the liver but is also up-regulated after LPS stimulation in macrophages and in the lungs of LPS-treated mice. Lipocalin 2 is an important factor in innate immune responses. Mice lacking Lipocalin 2 (mouse 24p3) develop normally but show hypersensitivity to bacterial infection. Lipocalin 2 binds to an iron siderophore that is required for the growth of bacteria. Lipocalin 2 is also implicated in kidney development and renal injury. In addition, Lipocalin 2 may be involved in tissue involution, a highly organized process of tissue restructuring. It was shown that Lipocalin 2 expression coincides with a high degree of apoptosis in an involuting tissue, Lipocalin 2 may promote the cell death of invading neutrophils and thus delay neutrophil entry into a tissue until the second phase of involution.A proapoptotic function for Lipocalin 2 has also been reported. When withdrawing IL-3 from in vitro cultured mouse cytokine-dependent hematopoietic cells, the transcription and secretion of Lipocalin 2 (mouse 24p3) is highly elevated and the conditioned medium (CM) derived from these cells causes apoptosis in certain hematopoietic cell types including primary hematopoietic cells. Importantly, high levels of Lipocalin 2 are associated with other types of cancers such as breast cancer.2 Human Lipocalin 2 (NGAL) was originally purified as a component complexed with metalloproteinase MMP-9, which is involved in the invasive behavior of many types of cancers. It was shown that the presence of Lipocalin 2 stabilizes MMP-9 activity. Importantly, it was shown that inhibition of MMP-9 activity reduces the invasive behavior in BCR-ABL + cells.3 Furthermore, it was reported that Lipocalin 2 is an adipokine with potential importance in insulin resistance associated with obesity.4
Lipocalin 2 (Lcn2)—also known as neutrophil gelatinase–associated Lipocalin, siderocalin, and 24p3. It was originally described as an acute-phase protein in the liver but is also up-regulated after LPS stimulation in macrophages and in the lungs of LPS-treated mice. Lipocalin 2 is an important factor in innate immune responses. Mice lacking Lipocalin 2 (mouse 24p3) develop normally but show hypersensitivity to bacterial infection. Lipocalin 2 binds to an iron siderophore that is required for the growth of bacteria. Lipocalin 2 is also implicated in kidney development and renal injury. In addition, Lipocalin 2 may be involved in tissue involution, a highly organized process of tissue restructuring. It was shown that Lipocalin 2 expression coincides with a high degree of apoptosis in an involuting tissue, Lipocalin 2 may promote the cell death of invading neutrophils and thus delay neutrophil entry into a tissue until the second phase of involution.A proapoptotic function for Lipocalin 2 has also been reported. When withdrawing IL-3 from in vitro cultured mouse cytokine-dependent hematopoietic cells, the transcription and secretion of Lipocalin 2 (mouse 24p3) is highly elevated and the conditioned medium (CM) derived from these cells causes apoptosis in certain hematopoietic cell types including primary hematopoietic cells. Importantly, high levels of Lipocalin 2 are associated with other types of cancers such as breast cancer.2 Human Lipocalin 2 (NGAL) was originally purified as a component complexed with metalloproteinase MMP-9, which is involved in the invasive behavior of many types of cancers. It was shown that the presence of Lipocalin 2 stabilizes MMP-9 activity. Importantly, it was shown that inhibition of MMP-9 activity reduces the invasive behavior in BCR-ABL + cells.3 Furthermore, it was reported that Lipocalin 2 is an adipokine with potential importance in insulin resistance associated with obesity.4
REFERENCES
1. Flower, D.R.: Biochem. J. 318:1–14, 1996
2. Yang, J. et al: Proc. Natl. Acad. Sci. USA 106:3913-8, 2009
3. leng, X. et al: Oncogene 27:6110-9, 2008
4. Yan, Q-W. et al: Diabetes 56:2533-40, 2007
2. Yang, J. et al: Proc. Natl. Acad. Sci. USA 106:3913-8, 2009
3. leng, X. et al: Oncogene 27:6110-9, 2008
4. Yan, Q-W. et al: Diabetes 56:2533-40, 2007
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CL0854 |
Target Protein Species: | Mouse |
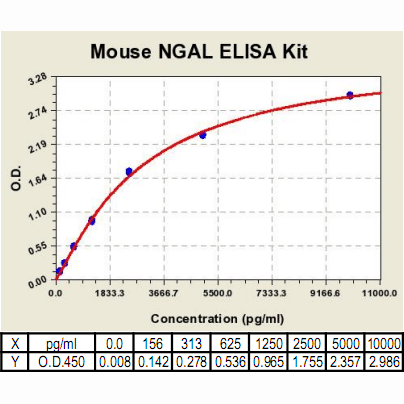
Range: | 156 pg/ml – 10000pg/ml |
Specificity: | No detectable cross-reactivity with other cytokines |
Storage: | Store at 4°C. Use within 6 months. |
ELISA Kits are based on standard sandwich enzyme-linked immunosorbent assay technology. Freshly prepared standards, samples, and solutions are recommended for best results.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой