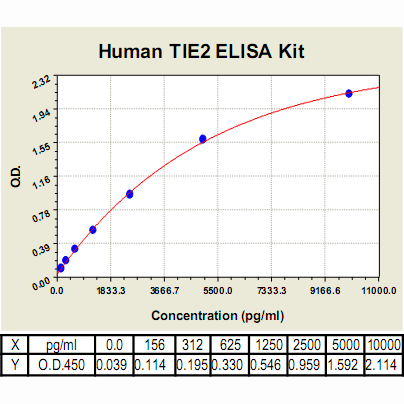
TIE2 ELISA Kit, Human |
 |
BACKGROUND Tie2 is a receptor tyrosine kinase (RTK) with a unique extracellular ligand-binding domain comprised of immunoglobulin (IG) domains, epithelial growth factor (EGF) repeats, and fibronectin-like 3 (FN3) repeats. It is highly conserved among vertebrate species from zebrafish to human, with the highest homology in the kinase domain. Tie2 is expressed on endothelial cells (ECs) and hematopoietic stem cells (HSCs).1 Tie2-/- mice die during embryogenesis at day 9.5 to 10.5; necropsy analyses have shown that the endothelial cells of such mice are present in normal numbers and assembled into tubes, but the vessels are immature and lack branch networks and hierarchical organization into large and small vessels. Observations in human subjects have revealed that deficient smooth muscle cell investment typical of venous malformations is associated with a mutation in the Tie2 RTK, suggesting that the Tie2 system may regulate the endothelial cell recruitment of stromal cells required to encase and thereby stabilize primitive endothelial tubes.2
The ligands of the Tie2 receptor have been identified as Ang1 and Ang2. Ang1 was identified as the major physiological ligand for Tie2, responsible for recruiting and sustaining periendothelial support cells. Ang2 was found to disrupt blood vessel formation in the developing embryo by antagonizing the effects of Ang1 and Tie2, and it was thus concluded that Ang2 represents a natural Ang1/Tie2 inhibitor. Extrapolation of these developmental findings to postnatal neovascularization has led to the dual inferences that Ang1 may induce maturation and stabilization of developing neovasculature, whereas Ang2 may cause destabilization required for additional sprout formation.3 Ang1 binds to Tie2 and induces its activation via tyrosine phosphorylation. Through the phosphatidylinositol 3-kinase (PI3K)-Akt pathway and others, Ang1 exerts prosurvival, antipermeability, and anti-inflammatory effects on endothelial cells (ECs). Furthermore, it was demonstrated that Ang2 possesses both partial agonistic as well as antagonistic action on Tie2 in ECs—alone, Ang2 is a weak but necessary activator of Tie2, whereas in the presence of Ang1, Ang2 inhibits Tie2 signaling. Moreover, ECs secrete Ang2, which in turn maintains a basal level of Tie2 phosphorylation.4
The ligands of the Tie2 receptor have been identified as Ang1 and Ang2. Ang1 was identified as the major physiological ligand for Tie2, responsible for recruiting and sustaining periendothelial support cells. Ang2 was found to disrupt blood vessel formation in the developing embryo by antagonizing the effects of Ang1 and Tie2, and it was thus concluded that Ang2 represents a natural Ang1/Tie2 inhibitor. Extrapolation of these developmental findings to postnatal neovascularization has led to the dual inferences that Ang1 may induce maturation and stabilization of developing neovasculature, whereas Ang2 may cause destabilization required for additional sprout formation.3 Ang1 binds to Tie2 and induces its activation via tyrosine phosphorylation. Through the phosphatidylinositol 3-kinase (PI3K)-Akt pathway and others, Ang1 exerts prosurvival, antipermeability, and anti-inflammatory effects on endothelial cells (ECs). Furthermore, it was demonstrated that Ang2 possesses both partial agonistic as well as antagonistic action on Tie2 in ECs—alone, Ang2 is a weak but necessary activator of Tie2, whereas in the presence of Ang1, Ang2 inhibits Tie2 signaling. Moreover, ECs secrete Ang2, which in turn maintains a basal level of Tie2 phosphorylation.4
REFERENCES
1. Hayes, A.J. et al: Microvascu. Res. 58:224-37, 1999
2. Vikkula, M. et al: Cell 87:1181-90, 1996
3. Maisonpierre,P.C. et al: Science 277:55-60, 1997
4. Yuan, H.T. et al: Mol. Cell. Biol. 29:2011-22, 2009
2. Vikkula, M. et al: Cell 87:1181-90, 1996
3. Maisonpierre,P.C. et al: Science 277:55-60, 1997
4. Yuan, H.T. et al: Mol. Cell. Biol. 29:2011-22, 2009
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CL0519 |
Target Protein Species: | Human |
Range: | 156pg/ml – 10000pg/ml |
Specificity: | Cross-reactivates with human TIE1< 0.1%. |
Storage: | Store at 4°C. Use within 6 months. |
ELISA Kits are based on standard sandwich enzyme-linked immunosorbent assay technology. Freshly prepared standards, samples, and solutions are recommended for best results.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой