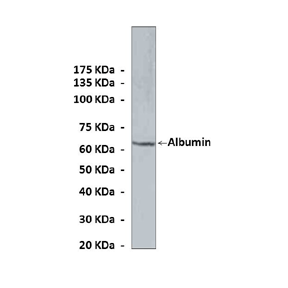
Anti-Albumin: Monoclonal Albumin Antibody |
 |
BACKGROUND Human serum albumin (HSA) is the most abundant protein in human blood plasma. It is synthesized in the liver as preproalbumin, and an N-terminal peptide is removed before the nascent protein is released from the rough endoplasmic reticulum. The product, proalbumin, is cleaved in the Golgi vesicles to produce the secreted albumin.1
Albumin is essential in maintaining the osmotic pressure for proper distribution of body fluids between intravascular compartments and body tissues. When ionized in water at physiological pH 7.4, Albumin is negatively charged. Albumin contains eleven distinct binding domains for hydrophobic compounds. One hemin and six long-chain fatty acids can bind to albumin at the same time. Albumin also acts as a plasma carrier by non-specifically binding several hydrophobic steroid hormones.2 The general structure of albumin is characterized by several long beta sheets, which allows it to maintain a relatively static shape, which is essential for regulating blood pressure.
Albumin, along with α1-acid glycoprotein (AGP), are plasma proteins that can act as drug carriers with drug pharmacokinetic implications. Albumin has an important clinical impact for drugs that have a relatively narrow therapeutic index.3
REFERENCES
REFERENCES
1. Emerson TE: Critical Care Med. 17:690-694, 1989.
2. Zunszain PA et al.: BMC Struct Biol. 3:6, 2003.
3. Fehske KJet al.: Mol. Pharmacol. 21: 387-393, 1982.
2. Zunszain PA et al.: BMC Struct Biol. 3:6, 2003.
3. Fehske KJet al.: Mol. Pharmacol. 21: 387-393, 1982.
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CC10022 |
Antigen: | Purified Human Serum Albumin protein. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Rat, Mouse, Cow |
Applications & Suggested starting dilutions: | WB 1:1000 IP 1:50 IHC 1:100 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 65 kDa |
Specificity/Sensitivity: | Detects endogenous levels of albumin protein in various cell lysates. |
Storage: | Store at 4° C for frequent use; at -20° C for at least one year. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой