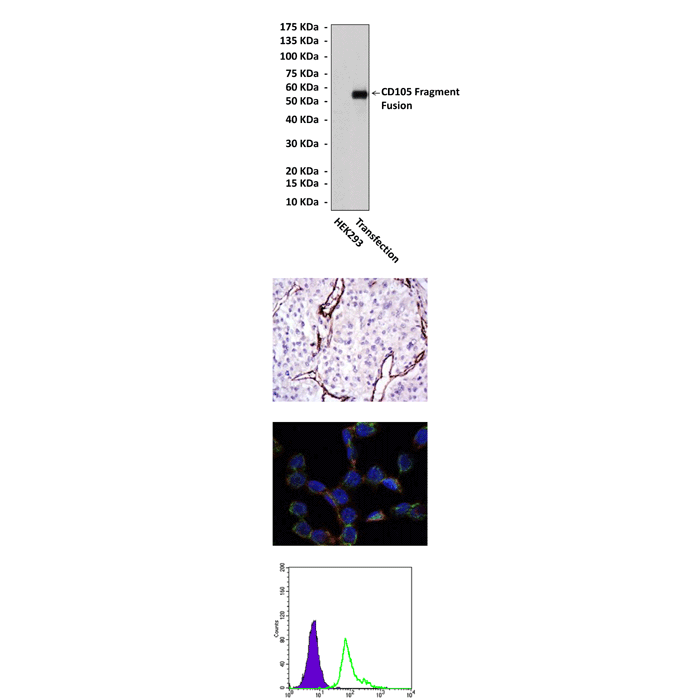
Anti-CD105: Mouse CD105 Antibody |
 |
BACKGROUND Endoglin (CD105) is a transmembrane glycoprotein expressed on activated vascular endothelial cells. It is an accessory protein of the transforming growth factor-beta (TGF-beta) receptor system and composed of two disulfide-linked subunits of 95 kDa each forming a 180-kDa homodimeric mature protein. Endoglin is expressed as two isoforms, designated long (L-CD105) and short (S-CD105), based on the length of the cytoplasmic domain. It is a marker of activated endothelium, and its vascular expression is limited to proliferating cells.1 Mutation of the endoglin gene is associated with hereditary hemorrhagic telangiectasias, or Osler-Weber-Rendu syndrome. In addition to its expression on endothelial cells, endoglin is found on the surface of several other cell types. The expression of endoglin is elevated on the endothelial cells of healing wounds, developing embryos, inflammatory tissues, and solid tumors. Several studies identified endoglin expression in several solid tumor types, with the level of expression correlating with various clinicopathologic factors including decreased survival and presence of metastases.2 Attempts to target endoglin and the cells that express this protein in tumor-bearing mice have yielded promising results.
Endoglin signaling is initiated by binding of TGF-beta and results in a series of activation steps leading to transcriptional activity. Endoglin binds several components of the TGF- superfamily including activin-A, BMP-7 and BMP-2. In particular, it binds TGF- 1 and - 3 with high affinity by associating with the TGF- type II receptor. TGF-beta ligand binds to a TbetaR-II homodimer, which then recruits the type I receptor ALK1 or ALK5 to form a heterotetrameric receptor complex. In endoglin-expressing cells, endoglin is included as a dimer in this receptor complex. TbetaR-II then phosphorylates either ALK1 or ALK5 in a highly conserved glycine/serine-rich cytoplasmic domain, causing a conformational change in the receptor. Signaling is propagated to the nucleus via phosphorylation of Smad proteins, which act as transcriptional coactivators or corepressors.3
Endoglin signaling is initiated by binding of TGF-beta and results in a series of activation steps leading to transcriptional activity. Endoglin binds several components of the TGF- superfamily including activin-A, BMP-7 and BMP-2. In particular, it binds TGF- 1 and - 3 with high affinity by associating with the TGF- type II receptor. TGF-beta ligand binds to a TbetaR-II homodimer, which then recruits the type I receptor ALK1 or ALK5 to form a heterotetrameric receptor complex. In endoglin-expressing cells, endoglin is included as a dimer in this receptor complex. TbetaR-II then phosphorylates either ALK1 or ALK5 in a highly conserved glycine/serine-rich cytoplasmic domain, causing a conformational change in the receptor. Signaling is propagated to the nucleus via phosphorylation of Smad proteins, which act as transcriptional coactivators or corepressors.3
REFERENCES
1. Letamendia, A. et al: J. Biol. Chem. 273:33011-9, 1998
2. Fonsatti, E. Et al: Oncogene 22:6557-63, 2003
3. lebrin, F. Et al: Cardiovasc. Res. 65:599–608, 2005
2. Fonsatti, E. Et al: Oncogene 22:6557-63, 2003
3. lebrin, F. Et al: Cardiovasc. Res. 65:599–608, 2005
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
|
Cat.No.:
|
CP10396
|
|
Antigen:
|
Recombinant human CD105 fragments expressed in E. coli.
|
|
Isotype:
|
Mouse IgG1
|
|
Species & predicted
species cross-
reactivity ( ):
|
Human
|
|
Applications &
Suggested starting
dilutions:*
|
WB 1:1000
IP n/d
IHC 1:50 - 1:200
ICC 1:50 - 1:200
FACS 1:50 - 1:200
|
|
Predicted Molecular
Weight of protein:
|
95 kDa
|
|
Specificity/Sensitivity:
|
Detects CD105 proteins in various cell lysate.
|
|
Storage:
|
Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles.
|
*Optimal working dilutions must be determined by end user.
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой