Anti-FOXD3: Mouse FOXD3 Antibody |
 |
BACKGROUND The winged-helix or forkhead class of transcription factors has been shown to play important roles in cell specification and lineage segregation. Foxd3 (previously published as Hfh2) is a member of forkhead-box (FOX) family transcription factors characterized by a monomeric DNA binding domain for nuclear localization and transcriptional regulation. Foxd3 was isolated in a low-stringency screen for forkhead family genes. Originally termed “Genesis,” Foxd3 was reported to be expressed only in mouse ES cells and their malignant equivalents. In addition, Foxd3 expression is one of several diagnostic markers characteristic of human ES cell lines. Foxd3 is required for maintenance of multipotency and self-renewal of embryonic stem (ES) cells and trophoblast stem cells in culture, and Foxd3 null mutant embryos do not survive past early gastrulation. However, Foxd3 expression is also detected during early embryogenesis in the epiblast and later in neural crest cells. Foxd3 is one of the initial markers of the NC lineage, and when removed from the NC, most NC progenitors and derivatives are markedly affected, both in vivo and in explant cultures. In these NC-specific Foxd3 mutant embryos, many of the bones and cartilage of the developing craniofacial skeleton are absent or markedly malformed, and the face does not fuse at the midline. The peripheral nervous system and enteric nervous system (ENS) are almost completely absent, and dorsal root ganglia are reduced in size, all because of defective maintenance of the NC progenitor pool within the premigratory and early migratory NC. Moreover, FOXD3 was found to be a stemness factor that prevents the production of melanocyte progenitors from the developing neural crest. FOXD3 controls the lineage choice between neural/glial and pigment cells by repressing MITF during the early phase of neural crest migration.1 Whereas, Foxd3 and Pax3 act together to affect survival and maintenance of cardiac NC progenitors, and loss of these progenitors catastrophically affects key aspects of later cardiovascular development.2 In addition, Foxd3 has also been implicated in the control of differentiation in multiple systems: Overexpression of Foxd3 in a myeloid cell line prevents appropriate maturation of these cells into granulocytes. Foxd3 mRNA can also induce the formation of mesoderm in Xenopus. It was also shown that Foxd3 is required for the maintenance of pluripotent cells in the preimplantation and peri-implantation stages of mouse embryogenesis.3 It was shown that FOXD3 levels are upregulated following attenuation of B-RAF and mitogen-activated protein/extracellular signal-regulated kinase (ERK) kinase (MEK) signaling in mutant B-RAF harboring human melanoma cells. FOXD3 is suppressed by B-RAF. Additionally, FOXD3-induced cell cycle arrest was prevented by p53 depletion and, to a lesser extent, p21Cip1 depletion.4 Furthermore, silencing of transcription factor FOXD3 is a key event early in leukemogenesis in both mouse and human chronic lymphocytic leukemia (CLL). It was shown that loss of Foxd3 expression due to an NF-κB p50/p50:HDAC1 repressor complex occurs in TCL1-positive B cells.5
REFERENCES
1. Thomsa, A.J. & Erickson, C.A.: 136:1849-58, 2009
2. Nelms, B.L. et al: Genesis49:10-23, 2011
3. Kos, R. Et al: Development 128:1467-79, 2001
4. Abel, E.V. & Aplin, A.E.: Cancer Res. 70:2891–900, 2010
5. Chen, S.S. et al: Proc. Natl. Acad. Sci. USA 106:13433-8, 2009
2. Nelms, B.L. et al: Genesis49:10-23, 2011
3. Kos, R. Et al: Development 128:1467-79, 2001
4. Abel, E.V. & Aplin, A.E.: Cancer Res. 70:2891–900, 2010
5. Chen, S.S. et al: Proc. Natl. Acad. Sci. USA 106:13433-8, 2009
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10347 |
Antigen: | Raised against recombinant human FOXD3 fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
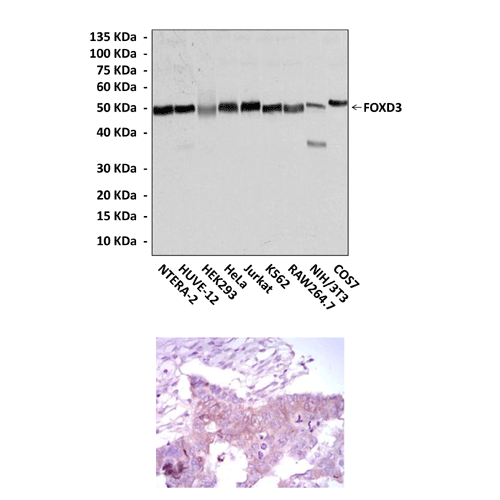
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:50 - 1:200 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 50 kDa |
Specificity/Sensitivity: | Detects endogenous FOXD3 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой