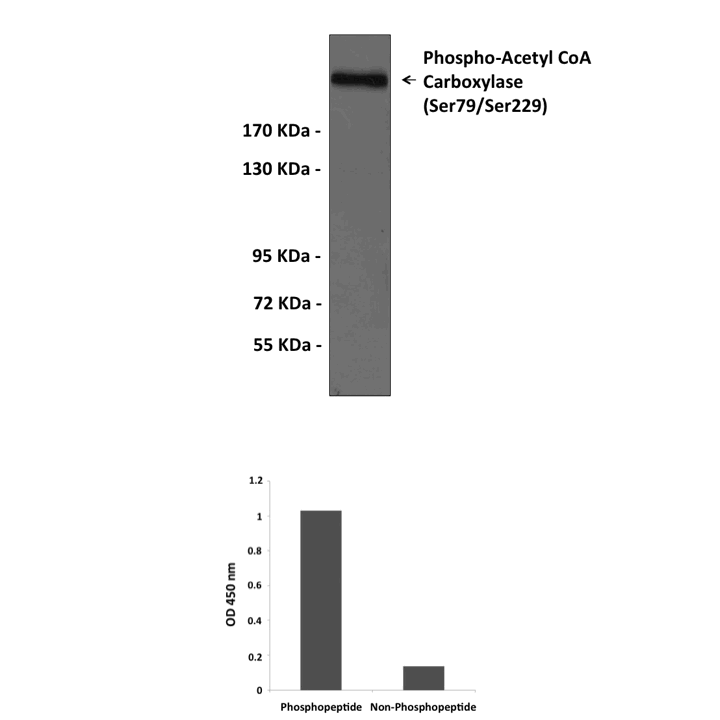
Anti-Phospho-ACC1/2: Rabbit Acetyl-CoA Carboxylase, Phospho-Ser79 (ACC1)/Ser221 (ACC2) Antibody |
 |
BACKGROUND Acetyl-CoA carboxylase (ACC or ACC1) catalyzes the biotin-dependent carboxylation of acetyl-CoA to produce malonyl-CoA. This is the first and the committed step in the biosynthesis of long-chain fatty acids. The most important function of ACC is to provide the malonyl-CoA substrate for the biosynthesis of fatty acids. The activity of ACC can be controlled at the transcriptional level as well as by small molecule modulators and covalent modification.1 A second isoform of ACC, ACC2, is associated with the mitochondrial membrane and produces malonyl-CoA that regulates fatty acid oxidation by potently inhibiting the carnitine palmitoyltransferases (CPT-Is). Mice that are deficient in ACC2 have elevated fatty acid oxidation and reduced body fat content and body weight, despite consuming more food.2 Therefore, inhibitors against ACCs might be efficacious for the treatment of obesity and diabetes (metabolic syndrome). The activity of the enzyme is also controlled by reversible phosphorylation. The enzyme is inhibited if phosphorylated; the phosphorylation can result when the hormones glucagon or epinephrine bind to their receptors, but the main cause of phosphorylation is due to a rise in AMP levels when the energy status of the cell is low, leading to the activation of the AMPK.3
The amino acid sequences of ACC1 and ACC2 had about 60 and 80% identity, respectively. Ser78 and Ser80, which were found to be critical for the phosphorylation and subsequent inactivation of rat ACC (Ser77 and Ser79 of rat ACC1), are conserved in ACC2 and are represented as Ser219 and Ser221, respectively. On the other hand, Ser1201, which is also a phosphorylation site in rat ACC1 (Ser1200 of rat ACC1), is not conserved in ACC2. AMPK phosphorylates ACC1 at multiple residues, although phosphorylation at a single serine Ser-80 in ACC1 accounts for the resulting inactivation. AMPK also inactivates ACC2 via phosphorylation at the site equivalent to Ser-80 on ACC1 (Ser-221 in human ACC2).4
REFERENCES
1. Tong L: Cell. Mol. Life Sci. 62:1784-1803, 2005.
2. Choi CS et al.: Proc. Natl. Acad. Sci. USA, 104: 16480-16485, 2007.
3. Janovska A et al.: Mol. and Cell. Endocrinol. 284:1-10, 2008.
4. Hadad, S.M. et al: BMC Cancer 9:307, 2007
2. Choi CS et al.: Proc. Natl. Acad. Sci. USA, 104: 16480-16485, 2007.
3. Janovska A et al.: Mol. and Cell. Endocrinol. 284:1-10, 2008.
4. Hadad, S.M. et al: BMC Cancer 9:307, 2007
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CG1001 |
Antigen: | Range AA65 to 95 |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Rat, Mouse |
Applications & Suggested starting dilutions:* | WB 1:500-1:1000 IP n/d IHC n/d ICC n/d FACS n/d ELISA 1:1000 |
Predicted Molecular Weight of protein: | 265 KDa |
Specificity/Sensitivity: | Detects endogenous ACC1/2 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
|
| ||||||||||
| ||||||||||
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой