Anti-Phospho-Akt: Rabbit Akt, Phospho-Ser473 Antibody |
 |
BACKGROUND AKT, also called protein kinase B (PKB), is a serine/threonine-specific protein kinase originally identified as the oncogene in the transforming retrovirus, AKT8. It plays an important role in mammalian cellular signaling.1 In humans, the Akt family has three members: Akt1, Akt2, and Akt3. Akt1 is involved in cellular survival pathways by inhibiting apoptotic processes. Akt1 is also able to induce protein synthesis pathways, and is therefore a key signaling protein in the cellular pathways that lead to skeletal muscle hypertrophy, and general tissue growth. Since it can block apoptosis, and thereby promote cell survival, Akt1 has been implicated as a major factor in many types of cancer.2 Akt2 is an important signaling molecule in the insulin signaling pathway. It is required to induce glucose transport.3 The role of Akt3 is less clear, though it appears to be predominantly expressed in the brain. It has been reported that mice lacking Akt3 have small brains.4
Each Akt isomer possesses an N-terminal pleckstrin homology (PH) domain (residues 1–113), a kinase domain (residues 150–408) that is similar to those found in other AGC members such as cAMP-dependent protein kinase and protein kinase C, and a C-terminal regulatory domain (residues 409–480) containing a hydrophobic motif. During the activation process, cytosolic Akt is recruited to the plasma membrane through the interaction of its PH domain with phosphatidylinositol 3,4,5-trisphosphate (PIP3), which is generated by phosphatidylinositol 3-kinase in response to the growth factor receptor stimulation. It is thought that the membrane interaction results in conformational changes of Akt, exposing a pair of residues, Thr308 at the activation loop of the kinase domain and Ser473 at the hydrophobic motif of the regulatory domain, for phosphorylation by the phosphoinositide-dependent kinase 1 (PDK1) and a putative kinase PDK2 (mTOR), respectively. It has been demonstrated that the phosphorylation at both Thr308 and Ser473 is required for the full activation of Akt.5 The phosphorylated active Akt in turn phosphorylates downstream effectors such as GSK3, PFK2, and BAD.
REFERENCES
1. Chan T et al.: Annu. Rev. Biochem. 68:965-1014, 1999.
2. Bussink J et al.: Lancet Oncol. 9:288-296, 2008.
3. Ng Y et al.: Cell Metabolism, 7: 348-356, 2008.
4. Easton RM et al.: Mol Cell Biol. 25: 1869–1878, 2005.
5. Balendran, A. et al: Curr. Biol. 9:393-404, 1999
2. Bussink J et al.: Lancet Oncol. 9:288-296, 2008.
3. Ng Y et al.: Cell Metabolism, 7: 348-356, 2008.
4. Easton RM et al.: Mol Cell Biol. 25: 1869–1878, 2005.
5. Balendran, A. et al: Curr. Biol. 9:393-404, 1999
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CG1002 |
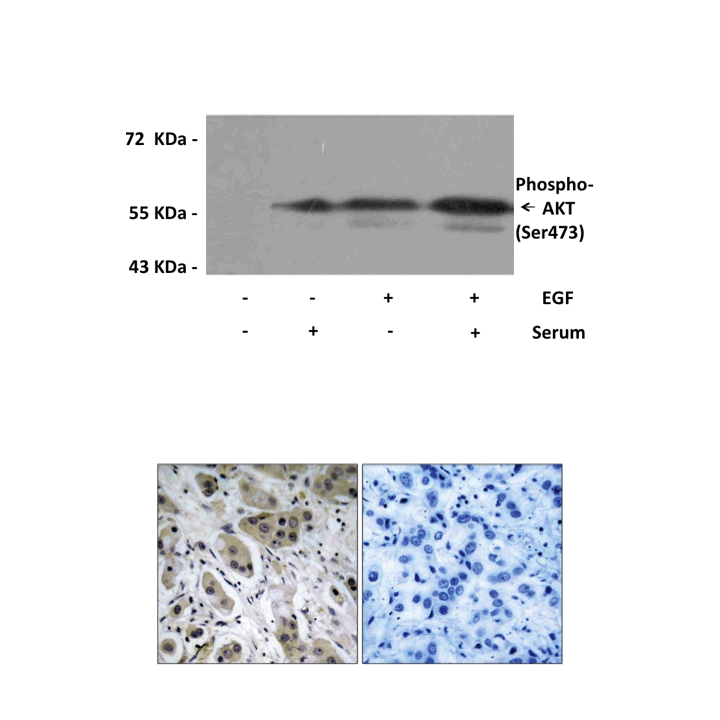
Antigen: | Peptide sequence around phosphorylation site of serine 473 (Q-F-S(p)-Y-S), according to the protein AKT1. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:500-1:1000 IP n/d IHC 1:50-1:100 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 60 KDa |
Specificity/Sensitivity: | Detects endogenous AKT (Ser473) proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой