Anti-COX4I1: Mouse COX4I1 Antibody |
 |
BACKGROUND Cytochrome c oxidase (EC 1.9.3.1) (COX) belongs to the superfamily of terminal oxidases present in all aerobic organisms. In eukaryotic cells, cytochrome c oxidase is embedded as a dimer in the mitochondrial inner membrane. Electron transfer by the enzyme from cytochrome c to molecular oxygen is coupled to proton pumping across the membrane, contributing to the mitochondrial membrane potential, resulting in a proton and charge gradient that is then employed by the F0F1-ATPase to synthesize ATP. The redox centers involved in electron transfer are two copper centers (CuA and CuB) and two heme A moieties (a and a3). The eukaryotic enzyme contains three mitochondrial DNA (mtDNA)-encoded subunits: MTCO1, MTCO2, and MTCO3. These subunits are homologous to polypeptides found in bacterial cytochrome c oxidases. They form both the catalytic and structural core of the enzyme. The hydrophobic interior of enzyme subunit 1 (MTCO1) coordinates the heme a group and a binuclear center composed of heme a3 and CuB. The intermembrane space domain of enzyme subunit 2 (MTCO2) coordinates the binuclear CuA center. In addition to the three mtDNA-encoded subunits, mammalian cytochrome c oxidase contains 10 nuclear DNA-encoded subunits: COX4, COX5A, COX5B, COX6A, COX6B, COX6C, COX7A, COX7B, COX7C, and COX8. In humans, COX6A and COX7A exist as skeletal/cardiac muscle (COX6A2 and COX7A1) and ubiquitously expressed (COX6A1 and COX7A2) isoforms. Although not confirmed at the protein level, human isoforms of COX4, COX6B, and COX8 have also been identified.1 Cytochrome c oxidase subunit 4 isoform 1 (COX4I1) is the nuclear-encoded subunit IV isoform 1 of the human mitochondrial respiratory chain enzyme. It is located at the 3' of the NOC4 (neighbor of COX4) gene in a head-to-head orientation, and shares a promoter with it.
Cytochrome c oxidase (COX) deficiencies are one of the most common defects of the respiratory chain found in mitochondrial diseases. cytochrome c oxidase (COX) biosynthesis requires numerous assembly factors that do not form part of the final complex but participate in prosthetic group synthesis and metal delivery in addition to membrane insertion and maturation of cytochrome c oxidase (COX) subunits. Human diseases associated with cytochrome c oxidase (COX) deficiency including encephalomyopathies, Leigh syndrome, hypertrophic cardiomyopathies, and fatal lactic acidosis are caused by mutations in cytochrome c oxidase (COX) subunits or assembly factors. Owing to the importance of the enzyme, pathogenetic mutations affecting cytochrome c oxidase (COX) frequently result in severe, often fatal metabolic disorders.2 In the last decade, numerous animal models have been created to understand the pathophysiology of cytochrome c oxidase (COX) deficiencies and the function of assembly factors. These animal models, ranging from invertebrates to mammals, in most cases mimic the pathological features of the human diseases.3
Cytochrome c oxidase (COX) deficiencies are one of the most common defects of the respiratory chain found in mitochondrial diseases. cytochrome c oxidase (COX) biosynthesis requires numerous assembly factors that do not form part of the final complex but participate in prosthetic group synthesis and metal delivery in addition to membrane insertion and maturation of cytochrome c oxidase (COX) subunits. Human diseases associated with cytochrome c oxidase (COX) deficiency including encephalomyopathies, Leigh syndrome, hypertrophic cardiomyopathies, and fatal lactic acidosis are caused by mutations in cytochrome c oxidase (COX) subunits or assembly factors. Owing to the importance of the enzyme, pathogenetic mutations affecting cytochrome c oxidase (COX) frequently result in severe, often fatal metabolic disorders.2 In the last decade, numerous animal models have been created to understand the pathophysiology of cytochrome c oxidase (COX) deficiencies and the function of assembly factors. These animal models, ranging from invertebrates to mammals, in most cases mimic the pathological features of the human diseases.3
REFERENCES
1. Capaldi, R.A.: Annu. Rev. Biochem. 59: 569-596, 1990
2. Pecina, P. et al: Physiol. Res. 53(Suppl. 1):S213-S223, 2004
3. Diaz, F.: Biochim Biophys Acta. 1802:100-10, 2010
2. Pecina, P. et al: Physiol. Res. 53(Suppl. 1):S213-S223, 2004
3. Diaz, F.: Biochim Biophys Acta. 1802:100-10, 2010
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10253 |
Antigen: | Purified recombinant human COX4I1 fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP 1:50 IHC n/d ICC 1:50 - 1:200 FACS 1:50 - 1:200 |
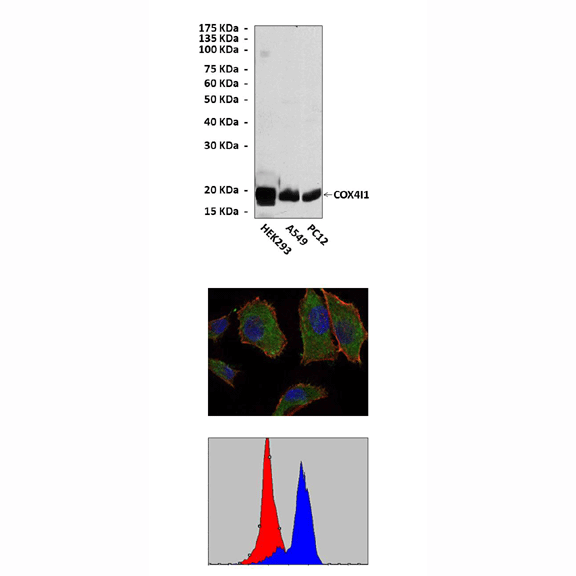
Predicted Molecular Weight of protein: | 19 kDa |
Specificity/Sensitivity: | Detects endogenous COX4I1 proteins without cross-reactivity with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой