Anti-Csk: Mouse C-Terminal Src Kinase Antibody |
 |
BACKGROUND The C-terminal src kinase (Csk) is a ubiquitously expressed, cytosolic enzyme capable of phosphorylating and inactivating several src-family protein tyrosine kinases (SFKs). It phosphorylates a consensus tyrosine near the C terminus of SFKs (Tyr527 in Src). This SFK carboxyl-terminal tyrosine is the only known physiological substrate of Csk. The phosphorylated SFK carboxyl-terminal tyrosine intramolecularly binds to the SH2 domain of SFKs. This interaction, known as Phospho-Tyr/SH2 interaction, together with binding between the SH2 kinase linker and the SH3 domain of SFKs (linker/SH3 interaction) stabilizes SFKs in a "closed" inactive conformation.1 It was demonstrated that Cbp (or called PAG), the Csk-binding protein, plays an important role in Csk-Src regulation. When Cbp is phosphorylated at a specific tyrosine residue (e.g., Y314 in rat), Csk binds to Cbp through its SH2 domain. The formation of the Csk–Cbp complex could boost the kinase activity of Csk and recruit Csk to phosphorylate Src.2 Because the Csk binding site could be phosphorylated by SFK, it has been suggested that there exists a feedback regulatory loop of SFK function.
In mammalian cells, there is another Csk member, CSK-homologous kinase (CHK). Both CSK and CHK are considered to be endogenous inhibitors of SFKs. Unlike CSK which is ubiquitously expressed in all mammalian tissues, whereas the expression of CHK is much more restricted; it is expressed predominantly in neurons and hemopoietic cells.3 They play pivotal roles in controlling cell adhesion, migration, and cancer progression.
In mammalian cells, there is another Csk member, CSK-homologous kinase (CHK). Both CSK and CHK are considered to be endogenous inhibitors of SFKs. Unlike CSK which is ubiquitously expressed in all mammalian tissues, whereas the expression of CHK is much more restricted; it is expressed predominantly in neurons and hemopoietic cells.3 They play pivotal roles in controlling cell adhesion, migration, and cancer progression.
REFERENCES
1. Chong YP et al.: J. Biol. Chem. 281:32988-99, 2006.
2. Jiang LQ et al.: Oncogene 25:5495-506, 2006.
3. Davidson D et al.: J. Biol. Chem. 272:1355-1362, 1997.
2. Jiang LQ et al.: Oncogene 25:5495-506, 2006.
3. Davidson D et al.: J. Biol. Chem. 272:1355-1362, 1997.
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10280 |
Antigen: | Raised against recombinant human Csk fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
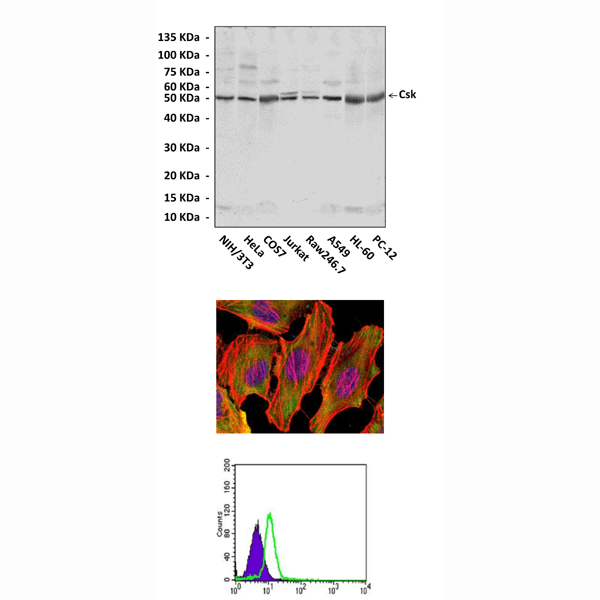
Applications & Suggested starting dilutions:* | WB 1:1000 IP 1:50 IHC n/d ICC 1:50 - 1:200 FACS 1:50 - 1:200 |
Predicted Molecular Weight of protein: | 50 kDa |
Specificity/Sensitivity: | Detects endogenous Csk proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой