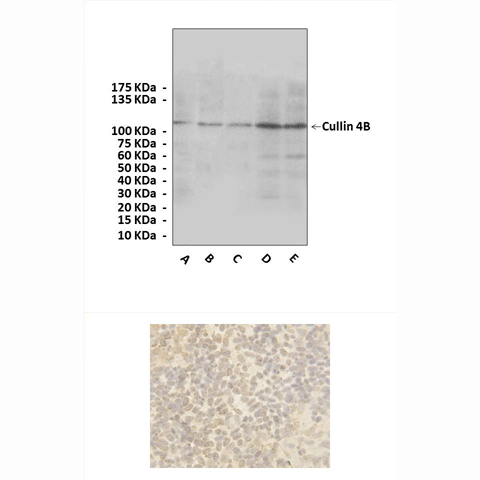
Anti-Cullin 4B: Rabbit Cullin 4B Antibody |
 |
BACKGROUND Cullins are a superfamily of ubiquitin ligases which are implicated in the regulation of several cellular functions. Cullins associate with ring proteins through their C-terminal domain, whereas the N-terminal region recruits a wide variety of receptor proteins that confer substrate specificity. They are modified by NEDD8 ubiquitin-like proteins, which stimulate their ubiquitin ligase activity. There are as many as six conserved members of the Cullin family of ubiquitin E3 ligases in mouse and human. Normally, a specific Cullin family member is associated with the SCF complex, which provides substrate specificity.1
In mammals, two closely related types of cullin, CUL4A and CUL4B have been identified. CUL4A and B encode subunits of E3-ubiquitin ligases implicated in diverse processes including nucleotide excision repair, regulating gene expression and controlling DNA replication fork licensing. CUL4B (∼104 kDa) is a paralog of CUL4A in humans (∼87 kDa), and the encoding genes map to Xq23 and 13q34, respectively. Alignment of the CUL4 sequences reveals that they are 83% identical. However, CUL4B has a unique NH2 terminus of 154 amino acids. Mutated CUL4B is the underlying defect in an X-linked mental retardation syndrome in which patients exhibit structural or functional abnormalities in the central nervous system, skeleton, and hematopoiesis. The overexpression of CUL4A is linked to breast and hepatocellular cancers. Studies on the role of CUL4B suggest that it works in cooperation with CUL4A to target certain proteins for degradation. It was shown that DDB1 is a substrate adaptor for both forms of CUL4. In addition to DDB2, other WD40-domain proteins have been identified which interact with DDB1 and serve as substrate receptor proteins for DDB1-CUL4–based E3 ligases.2 Recent studies have solidified the notion of DDB1-CUL4-RBX1 as a central platform for building E3s involved in various aspects of the UV-damage response, most importantly, cell cycle regulation and NER. In addition, it was demonstrated that Cul4B function is essential for beta-catenin degradation in both Drosophila and mouse.3 Furthermore mutations in CUL4B were identified in humans associated with mental retardation, relative macrocephaly, tremor and a peripheral neuropathy.4
In mammals, two closely related types of cullin, CUL4A and CUL4B have been identified. CUL4A and B encode subunits of E3-ubiquitin ligases implicated in diverse processes including nucleotide excision repair, regulating gene expression and controlling DNA replication fork licensing. CUL4B (∼104 kDa) is a paralog of CUL4A in humans (∼87 kDa), and the encoding genes map to Xq23 and 13q34, respectively. Alignment of the CUL4 sequences reveals that they are 83% identical. However, CUL4B has a unique NH2 terminus of 154 amino acids. Mutated CUL4B is the underlying defect in an X-linked mental retardation syndrome in which patients exhibit structural or functional abnormalities in the central nervous system, skeleton, and hematopoiesis. The overexpression of CUL4A is linked to breast and hepatocellular cancers. Studies on the role of CUL4B suggest that it works in cooperation with CUL4A to target certain proteins for degradation. It was shown that DDB1 is a substrate adaptor for both forms of CUL4. In addition to DDB2, other WD40-domain proteins have been identified which interact with DDB1 and serve as substrate receptor proteins for DDB1-CUL4–based E3 ligases.2 Recent studies have solidified the notion of DDB1-CUL4-RBX1 as a central platform for building E3s involved in various aspects of the UV-damage response, most importantly, cell cycle regulation and NER. In addition, it was demonstrated that Cul4B function is essential for beta-catenin degradation in both Drosophila and mouse.3 Furthermore mutations in CUL4B were identified in humans associated with mental retardation, relative macrocephaly, tremor and a peripheral neuropathy.4
REFERENCES
1. Petroski, M.D. & Deshaies, R.J.: Nat. Rev. Mol. Cell. Biol. 6:9-20, 2005
2. Guerrero-Santoro, J. et al: Cancer Res. 68:5014-22, 2008
3. Tripathi, R. et al: J. Biosci. 32:1133-8, 2007
4. Kerzendorfer, C. Et al: Hum. Mol. Genet. 19:1324-34, 2010
2. Guerrero-Santoro, J. et al: Cancer Res. 68:5014-22, 2008
3. Tripathi, R. et al: J. Biosci. 32:1133-8, 2007
4. Kerzendorfer, C. Et al: Hum. Mol. Genet. 19:1324-34, 2010
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CA1233 |
Antigen: | Short peptide from human Cullin 4B sequence. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:50 - 1:200 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 102 kDa |
Specificity/Sensitivity: | Detects endogenous levels of Cullin 4B proteins without cross-reactivity with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой