Anti-IRAK1: Polyclonal Interleulin-1 Receptor Activated Kinase Antibody |
 |
BACKGROUND Interleukin-1 receptor activated kinases (IRAKs) are key mediators in the signaling pathways of TLRs/IL-1Rs. By means of their kinase and adaptor functions, IRAKs initiate a cascade of signaling events eventually leading to induction of inflammatory target gene expression. Due to this pivotal role, IRAK function is also highly regulated via multiple mechanisms. Four IRAK members have been identified in the human genome (IRAK-1, 2, M, and 4), which seem to play distinct roles.1 IRAK-1 is a highly specific signaling molecule activated by the Toll/IL-1 receptor family. It is a multidomain protein consisting of an N-terminal death domain, a regulatory ProST (proline-, serine-, threonine-rich) region, a conventional serine/threonine protein kinase domain and a large C-terminal rest. After activation of TLR4 or the type 1 IL-1 receptor, MyD88 and IRAK1 are recruited to the activated receptor complex. IRAK1 becomes phosphorylated, dissociates from the receptor complex, and associates with TNF receptor-associated factor 6 (TRAF6). The signal is then distributed to multiple downstream targets, including NF-kB, c-Jun NH2-terminal kinase (JNK), and p38-alpha MAPK.2 The signaling function of IRAK-1 is independent of its enzymatic activity. The kinase domain and the ProST region serve as a self-activating kinetic switch module in IRAK-1. Two different phosphorylation events take place in a sequential fashion in IRAK-1.3 The first is an autophosphorylation of the kinase domain resulting in full enzymatic activity. This is followed by multiple phosphorylations in the ProST region. The introduction of negative charges adjacent to the death domain has two consequences: First hyperphosphorylated IRAK-1 dissociates from the upstream adapter MyD88 and thus leaves the active receptor complex allowing optimal interaction with the downstream adapter TRAF6. Second, hyperphosphorylated IRAK-1 is targeted to the proteasome where it is proteolytically degraded and thus removed out of the signaling chain. This self-limitation guarantees a transient IL-1 signal. This regulatory function is completely dispensable for IRAK-1's adapter function as a signaling molecule.
REFERENCES
1. Gottipati S et al.: Cell Signal. 20:269-276, 2008.
2. Akira S & Takeda K.: Nature Rev. Immunol. 4:499-511, 2004.
3. Neumann D et al.: J. Leuk. Biol. 84:807-813, 2008.
2. Akira S & Takeda K.: Nature Rev. Immunol. 4:499-511, 2004.
3. Neumann D et al.: J. Leuk. Biol. 84:807-813, 2008.
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CC1048 |
Antigen: | Sequence near the human IRAK1 carboxyl terminal. |
Isotype: | Affinity purified Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions: | WB 1:1000 IP n/d IHC (Paraffin) n/d ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 80 kDa |
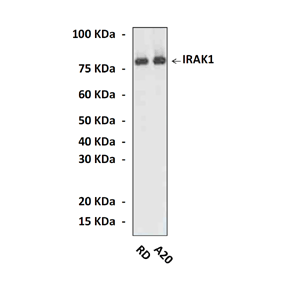
Specificity/Sensitivity: | Detects endogenous IRAK1 proteins in various cell lysates. |
Storage: | Store at 4° C for frequent use; at -20° C for at least one year. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой