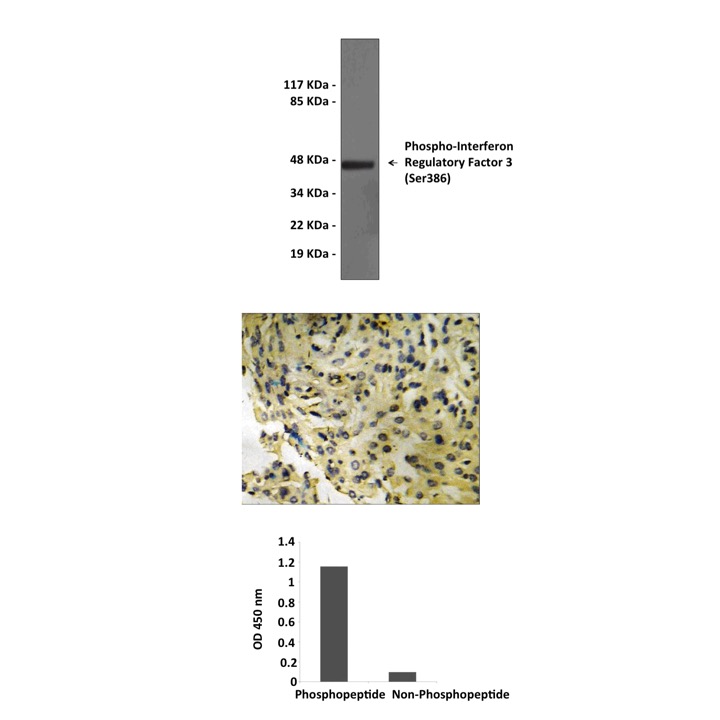
Anti-Phospho-IRF3: Rabbit Interferon Regulatory Factor 3, Phospho-Ser386 Antibody |
 |
BACKGROUND The interferon (IFN) regulatory factors (IRF) consist of a growing family of related transcription proteins. This family includes nine mammalian members, IRF-1 to IRF-9, as well as several viral homologs. All these proteins are characterized by a well-conserved N-terminal DNA-binding domain of about 120 amino acids, which recognizes similar DNA sequences (the consensus being 5'-AANNGAAA-3'), termed IRF-binding element/IFN-stimulated response element (ISRE). This binding element is present in the promoter of the IFN- / genes and of many IFN-stimulated genes. IRF-3 was originally identified as a member of the IRF family based on homology with other IRF family members and on binding to the ISRE of the IFN-stimulated gene 15 (ISG15) promoter. In addition, IRF-3 contains a C-terminal region with homology to the SMAD family of transcription factors. Several recent studies have focused attention on the unique molecular properties of IRF3 and its role in the regulation of IFN gene expression.1 IRF-3 is expressed constitutively in a variety of tissues, and the relative levels of IRF-3 mRNA do not change in virus-infected or IFN-treated cells. Following virus infection, IRF-3 is posttranslationally modified by protein phosphorylation at multiple serine and threonine residues, located in the carboxy-terminus of IRF-3.2 Phosphorylation is required for the cytoplasmic to nuclear translocation of IRF-3, its dimerization, stimulation of DNA binding and increased transcriptional activation, mediated through the association of IRF-3 with the CBP/p300 coactivator. Ser-386 is the target of the IRF-3 kinase and critical determinant for the activation of IRF-3.3 IRF-3 does not have any transcription activation domains.
It was shown that there are several signaling pathways leading to IRF-3 activation. Virus infection activates an uncharacterized VAK responsible for the C-terminal phosphorylation and subsequent activation of interferon regulatory factor 3 (IRF-3). It was shown that IKK-related kinases—IKKsigma/TBK-1—are components of VAK that mediate IRF-3 and IRF-7 phosphorylation and thus functionally link the NF-κB and IRF pathways in the development of the antiviral response.4 Virus-mediated activation of IRF-3 through VAK is dependent on viral entry and transcription, since replication deficient virus failed to induce IRF-3 activity. Recently, the range of inducers responsible for IRF-3 activation has increased. In addition to virus infection, recognition of bacterial infection mediated through lipopolysaccharide by Toll-like receptor 4 has also been reported. Furthermore, MAP kinase kinase kinase (MAP KKK)-related pathways and DNA-PK induce N-terminal phosphorylation of IRF-3.5
REFERENCES
1. Panne,D. et al: EMBO J. 23:4384-93, 2004
2. Lin, R. et al: Mol. Cell. Biol. 18:2986-96, 1998
3. Mori, M. et al: J. Biol. Chem. 279:9698-9702, 2004
4. Hiscott, J. et al: Annals New Yok Acad. Sci 1010:237-48, 2006
5. Servant, M.J. et al: Biochem. Pharmacol. 64:985-92, 2002
2. Lin, R. et al: Mol. Cell. Biol. 18:2986-96, 1998
3. Mori, M. et al: J. Biol. Chem. 279:9698-9702, 2004
4. Hiscott, J. et al: Annals New Yok Acad. Sci 1010:237-48, 2006
5. Servant, M.J. et al: Biochem. Pharmacol. 64:985-92, 2002
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CG1259 |
Antigen: | Range AA371 to 401 |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:500-1:1000 IP n/d IHC 1:50-1:100 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 47 kDa |
Specificity/Sensitivity: | Detects endogenous IRF3 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой