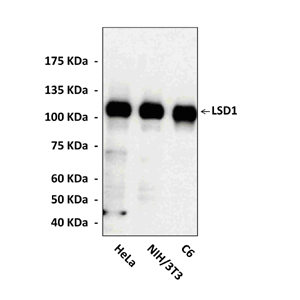
Anti-LSD1: Monoclonal Lysine-Specific Demethylase 1 Antibody (2H5) |
 |
BACKGROUND Lysine-specific demethylase 1 (LSD1; also known as BHC110) is a flavin-dependent monoamine oxidase, which can demethylate mono- and di-methylated lysines, specifically histone 3, lysines 4 and 9 (H3K4 and H3K9).1 Through a FAD-dependent oxidative reaction, LSD1 speficially removes histone H3K4me2 to H3K4me1 or H3K4me0. When forming a complex with androgen receptor (and possibly other nuclear hormone receptors), LSD1 changes its substrates to H3K9me2. LSD1 relieves repressive histone marks by demethylation of histone H3 at lysine 9 (H3-K9), thereby leading to de-repression of androgen receptor target genes.2 Functions of other components in LSD1 complex, such as CoREST and BHC80, as well as p53, have been further studied.3, 4 It's now known LSD1 complex mediates a coordinated histone modification switch through enzymatic activities as well as histone modification readers in the complex.
REFERENCES
1. Forneris, F. et al: Trends Biochem Sci.33:181, 2008.
2. Culhane, J.C. & Cole, P. A.: Curr Opin Chem Biol. 11:561, 2007.
3. Urbanucci, A. et al: BMC Cancer. 8:219, 2008.
4. Tsai, W.W. et al: Mol. Cell. Biol. 28:5139, 2008.
2. Culhane, J.C. & Cole, P. A.: Curr Opin Chem Biol. 11:561, 2007.
3. Urbanucci, A. et al: BMC Cancer. 8:219, 2008.
4. Tsai, W.W. et al: Mol. Cell. Biol. 28:5139, 2008.
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CB18672 |
Antigen: | E. coli-expressed recombinant human LSD1 protein fragments. |
Isotype: | Mouse monoclonal IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions: | WB 1:1000 IP n/d IHC (Paraffin) n/d ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 110 kDa |
Specificity/Sensitivity: | Specifically detects endogenous levels of LSD1 proteins. Does not cross-react with other related proteins. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой