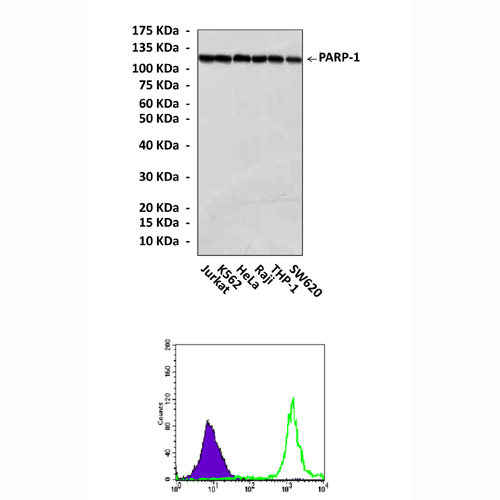
Anti-PARP-1: Mouse PARP-1 Antibody |
 |
BACKGROUND Poly (ADP-ribose) polymerase (PARP) catalyzes the post-translational modification of proteins by the addition of multiple ADP-ribose moieties. PARP transfers ADP-ribose from nicotinamide dinucleotide (NAD) to glu/asp residues on the substrate protein, and also polymerizes ADP-ribose to form long/branched chain polymers. PARP-1, one of 5 confirmed PARPs, is the most abundant and highly expressed enzyme.1 PARP-1 detects and relocates to single strand breaks or nicks in chromosomal DNA. PARP-1 is thought to play an important role in the initiation of the DNA repair pathway, although high levels of activation are also associated with increased apoptosis in response to genotoxic stress. In addition, PARP-1 may also operate downstream of the Raf-MEK-ERK pathway through direct interaction with ERK2 in the nucleus in a mechanism DNA damage.2 PARP inhibitors are being developed for use in a number of pathologies including cancer, diabetes, stroke and cardiovascular disease.
During cell death by apoptosis cleavage of PARP1 by caspases produces two fragments. The 25 kDa N-terminal peptide retains the DNA binding domains of PARP1. A C-terminal 85 kDa fragment has reduced catalytic activity and its detection is usually taken as a sensitive assay for apoptosis.3
During cell death by apoptosis cleavage of PARP1 by caspases produces two fragments. The 25 kDa N-terminal peptide retains the DNA binding domains of PARP1. A C-terminal 85 kDa fragment has reduced catalytic activity and its detection is usually taken as a sensitive assay for apoptosis.3
REFERENCES
1. Tong, W.M. et al: Biochem. Biophy. Acta 1552:27-37, 2001
2. Kim, M.Y. et al: Gene Dev. 19:1951-1967, 2005
3. Garnier, P. et al: J. Neurosci. 23:7967-73, 2003
2. Kim, M.Y. et al: Gene Dev. 19:1951-1967, 2005
3. Garnier, P. et al: J. Neurosci. 23:7967-73, 2003
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10319 |
Antigen: | Raised against recombinant human PARP-1 fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP 1:50 IHC n/d ICC n/d FACS 1:50 - 1:200 |
Predicted Molecular Weight of protein: | 116 kDa |
Specificity/Sensitivity: | Detects endogenous PARP-1 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой