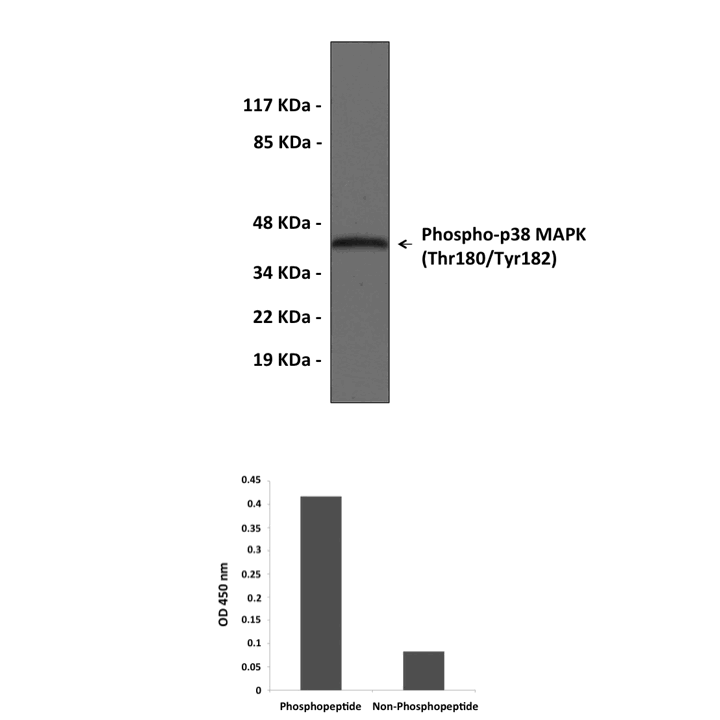
Anti-Phospho-p38 MAPK: Rabbit p38 MAPK, Phospho-Thr180/Tyr182 Antibody |
 |
BACKGROUND MAPK cascades are important mediators of the cellular response to a wide variety of extracellular signals, including mitogens, growth factors, cytokines, and cellular stress. There are 14 MAPKs in mammalian cells and these can be divided into four groups, the classical MAPKs (ERK 1 and ERK2), JNKs, p38s, and atypical MAPKs such as ERK3, ERK5, and ERK8.1
In mammalian cells, four isoforms of the p38 MAPKs have been described, p38alpha, p38beta, p38gamma, and p38delta. Of these, p38 and p38beta are ubiquitously expressed while p38gamma and p38delta are differentially expressed depending on tissue type. The p38 MAPKs are activated primarily in response to proinflammatory cytokines and cellular stress such as UV irradiation and osmotic and chemical shock. Activation of p38 occurs by dual phosphorylation of a Thr-Xaa-Tyr motif (Thr180 and Tyr182 in p38 MAPK-alpha) in the p38 kinase domain by an upstream MAPK kinase (MKK). Using MKK knockouts, it has been shown that in response to most stimuli MKK3 and MKK6 are the main MKKs activating p38alpha, possibly other isoforms, although in some circumstances, such as UV stimulation, MKK4 may also play a role. MKK6 and MKK3 are in turn activated by phosphorylation by a MAPK kinase kinase (MKKK). The MKKK responsible for activating the p38 cascade appears to be cell type and stimulus specific, and several MKKKs have been implicated in the activation of p38. These MKKKs include MLKs, Ask, and Tak.2
p38 isoforms have been implicated in several processes including cell survival, apoptosis, and immune function . These p38 MAPK isoforms appear to mediate distinct functions in vivo. It was shown that one subgroup (p38alpha and p38beta) is inhibited by a class of pyridinyl imidazole drugs, while the other subgroup (p38gamma and p38delta) is insensitive to these drugs. These p38 MAPKs phosphorylate both a common group of substrates and distinct substrates and they can be activated selectively by some extracellular stimuli, suggesting that they may exert distinct biological actions. Indeed, studies of HeLa cells indicate that while p38alpha induces apoptosis, p38beta2 promotes cell survival. These studies suggest that different p38 isoforms have overlapping, but also distinct physiological roles. The specificity of p38 MAPK signaling may be due to differences in substrate phosphorylation by individual p38 MAPKs and also to selective activation by MAPK kinases (MAPKKs). In addition, other two mechanisms may also be accounted for it. One mechanism of specificity is the selective formation of functional complexes between MAPKK and different p38 MAPKs. The formation of these complexes requires the presence of a MAPK docking site in the N-terminus of the MAPKK. The second mechanism that confers signaling specificity is the selective recognition of the activation loop (T-loop) of p38 MAPK isoforms.3
REFERENCES
1. Thomas, G.M. & Huganir, R.L.: Nature Rev. Neurosci. 5:173-83, 2004
2. Zarubin, T. & Han, J.: Cell Res. 15:11-18, 2005
3. Enslen, H. et al: EMBO J.19:1301-11, 2000
2. Zarubin, T. & Han, J.: Cell Res. 15:11-18, 2005
3. Enslen, H. et al: EMBO J.19:1301-11, 2000
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CG1383 |
Antigen: | Synthetic phosphopeptide derived from the region of p38 that contains threonine180 and tyrosine182 based on the human sequence. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:500-1:1000 IP n/d IHC n/d ICC n/d FACS n/d ELISA 1:5000 |
Predicted Molecular Weight of protein: | 41 kDa |
Specificity/Sensitivity: | Detects endogenous phosphorylated p38 MAPK (Thr180/Tyr182) proteins of all isoforms without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой