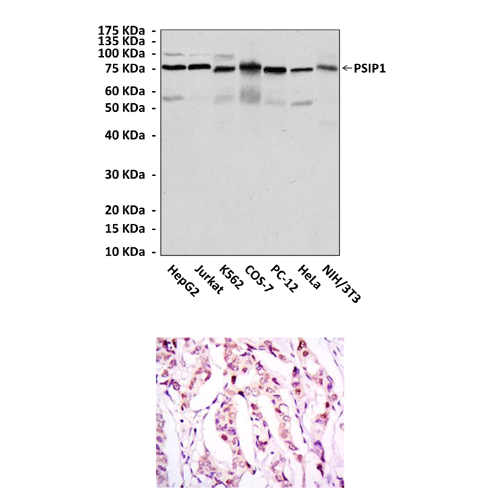
Anti-PSIP1/LEDGF/p75: Mouse PSIP1/LEDGF/p75 Antibody |
 |
BACKGROUND PC4- and SF2-interacting protein 1 (PSIP1) has been isolated in a number of independent experimental settings and has been assigned a variety of names and putative functions. Psip1 encodes two protein isoforms with molecular masses of 52 and 75 kDa. p52 and p75 were identified as interacting with transcription factor PC4 and shown to act as transcriptional coactivators. p75 is also referred to as lens epithelium-derived growth factor (LEDGF) since it was identified as a cell survival factor under a variety of conditions of environmental stress, and it has been implicated as a transcriptional regulator of stress-related genes . LEDGF/PSIP1/p75 is also a nuclear autoantigen targeted by autoantibodies in some patients with atopic dermatitis and other inflammatory conditions involving dysregulated apoptosis. It is thought to exert an antiapoptotic effect by transcriptional activation of stress-related genes, and it is cleaved by caspases.1
LEDGF/PSIP1/p75 has been reported to localize to chromatin in both interphase and mitotic chromosomes. The N-terminal domain of Psip1 contains a PWWP domain, a member of the Tudor domain "royal family" that includes chromo domains—some of which are known to bind to chromatin. The LEDGF/PSIP1/p75 PWWP domain affects the interaction of the protein with chromatin in vivo. LEDGF/PSIP1/p75 also contains AT hook-like motifs , and some of these have been implicated in DNA binding in vitro. It has been demonstrated that LEDGF/PSIP1/p75 is the cellular binding partner of lentiviral integrase (IN) proteins. LEDGF/PSIP1/p75 accounts for the characteristic propensity of Lentivirus to integrate within active transcription units and is required for efficient viral replication. In LEDGF/PSIP1/p75-depleted cell lines, reductions were seen in lentiviral infectivity compared to controls. The frequency of integration in transcription units was correlated with the cell-type specific levels of LEDGF/PSIP1/p75 expression.2 It has been demonstrated that LEDGF/PSIP1/p75 tightly associates with chromatin and has been implicated in regulation of cellular gene expression, epigenetic chromatin modifications and apoptosis. The host factor directly binds HIV-1, HIV-2, as well as other lentiviral INs and dramatically stimulates their strand transfer activity. LEDGF/PSIP1/p75 tethers lentiviral IN to host chromatin in the nucleus and plays a critical role in directing PICs to active genes during integration. LEDGF/PSIP1/p75 contains a pair of small structural domains: an ~92 residue PWWP domain at its N-terminus, responsible for binding to an as yet unidentified component of chromatin, and the IN binding domain (IBD, residues 347–429) within its C-terminal portion. The CCD and NTD of IN were both implicated in LEDGF/PSIP1/p75 binding: while the CCD is minimally sufficient, the NTD is required for high affinity binding. Deletion of the HIV-1 IN NTD, or a mutation destabilizing zinc coordination within this domain (His-12 to Asn), greatly reduced the interaction with LEDGF/PSIP1/p75. A close homolog of LEDGF/PSIP1/p75, hepatoma derived growth factor-related protein 2 (HRP2), contains conserved PWWP and IBD-like domains. Although HRP2 is able to interact with HIV-1 IN and stimulate its enzymatic activity in vitro, it remains to be established whether it plays a role in lentiviral integration. The structure of the LEDGF IBD, composed of a pair of α-helical hairpins, has been determined both separately and in complex with the HIV-1 IN CCD and NTD. This part of the protein-protein interface is essential for high affinity binding and stimulation of concerted DNA integration, and allows designs of complementary pairs of IN and LEDGF mutants for practical uses in gene therapy.3 Furthermore, it was shown that regulation of LEDGF/PSIP1/p75 expression was shown to be involved in a complex interplay mechanism between yet undefined regulatory elements to modulate gene expression.4
LEDGF/PSIP1/p75 has been reported to localize to chromatin in both interphase and mitotic chromosomes. The N-terminal domain of Psip1 contains a PWWP domain, a member of the Tudor domain "royal family" that includes chromo domains—some of which are known to bind to chromatin. The LEDGF/PSIP1/p75 PWWP domain affects the interaction of the protein with chromatin in vivo. LEDGF/PSIP1/p75 also contains AT hook-like motifs , and some of these have been implicated in DNA binding in vitro. It has been demonstrated that LEDGF/PSIP1/p75 is the cellular binding partner of lentiviral integrase (IN) proteins. LEDGF/PSIP1/p75 accounts for the characteristic propensity of Lentivirus to integrate within active transcription units and is required for efficient viral replication. In LEDGF/PSIP1/p75-depleted cell lines, reductions were seen in lentiviral infectivity compared to controls. The frequency of integration in transcription units was correlated with the cell-type specific levels of LEDGF/PSIP1/p75 expression.2 It has been demonstrated that LEDGF/PSIP1/p75 tightly associates with chromatin and has been implicated in regulation of cellular gene expression, epigenetic chromatin modifications and apoptosis. The host factor directly binds HIV-1, HIV-2, as well as other lentiviral INs and dramatically stimulates their strand transfer activity. LEDGF/PSIP1/p75 tethers lentiviral IN to host chromatin in the nucleus and plays a critical role in directing PICs to active genes during integration. LEDGF/PSIP1/p75 contains a pair of small structural domains: an ~92 residue PWWP domain at its N-terminus, responsible for binding to an as yet unidentified component of chromatin, and the IN binding domain (IBD, residues 347–429) within its C-terminal portion. The CCD and NTD of IN were both implicated in LEDGF/PSIP1/p75 binding: while the CCD is minimally sufficient, the NTD is required for high affinity binding. Deletion of the HIV-1 IN NTD, or a mutation destabilizing zinc coordination within this domain (His-12 to Asn), greatly reduced the interaction with LEDGF/PSIP1/p75. A close homolog of LEDGF/PSIP1/p75, hepatoma derived growth factor-related protein 2 (HRP2), contains conserved PWWP and IBD-like domains. Although HRP2 is able to interact with HIV-1 IN and stimulate its enzymatic activity in vitro, it remains to be established whether it plays a role in lentiviral integration. The structure of the LEDGF IBD, composed of a pair of α-helical hairpins, has been determined both separately and in complex with the HIV-1 IN CCD and NTD. This part of the protein-protein interface is essential for high affinity binding and stimulation of concerted DNA integration, and allows designs of complementary pairs of IN and LEDGF mutants for practical uses in gene therapy.3 Furthermore, it was shown that regulation of LEDGF/PSIP1/p75 expression was shown to be involved in a complex interplay mechanism between yet undefined regulatory elements to modulate gene expression.4
REFERENCES
1. Sutherland, H.G. et al: Mol. Cell. Biol. 26:7201-10, 2006
2. Marshall, H.M. et al: PLoS ONE 2:e1340, 2007
3. Hare, S. et al: PLoS Pathog. 5:e1000259, 2009
4. Desfarges, S. et al: Retrovirol. 6:O20, 2009
2. Marshall, H.M. et al: PLoS ONE 2:e1340, 2007
3. Hare, S. et al: PLoS Pathog. 5:e1000259, 2009
4. Desfarges, S. et al: Retrovirol. 6:O20, 2009
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10364 |
Antigen: | Raised against recombinant human PSIP1 fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:50 - 1:200 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 75 kDa |
Specificity/Sensitivity: | Detects PSIP1 proteins in various cell lysate. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой