Anti-PTK6/Brk: Mouse PTK6/Brkl Antibody |
 |
BACKGROUND PTK6 (also known as Brk) with its murine homolog being called Sik, is an intracellular tyrosine kinase that is distantly related to Src family tyrosine kinases. Members of the PTK6 family are defined by a highly conserved exon structure that is distinct from other major intracellular tyrosine kinase families. Like Src family kinases, PTK6 contains SH3, SH2, and tyrosine kinase catalytic (Kinase) domains and it is negatively regulated by phosphorylation of a C-terminal tyrosine residue. The SH3 domain of PTK6 interacts with the N-terminal half of the linker (Linker) region between the SH2 and Kinase domains to maintain the protein in an inactive conformation along with the phosphorylated Tyr447-SH2 interaction.1 Unlike Src family kinases, PTK6 is not myristoylated or specifically targeted to the membrane. As a consequence, PTK6 has been localized to different cellular compartments, including the nucleus, where it might have a distinct set of substrates and interacting proteins. It was demonstrated that recruitment of PTK6 to the plasma membrane is required for its oncogenic function. Localization of PTK6 to the plasma membrane enhanced the ability of PTK6 to promote proliferation, cell survival and migration and to permit anchorage-independent colony formation.2
PTK6 is strongly expressed in a large subset of breast carcinomas, and some metastatic melanomas, moderately overexpressed in some colon cancers, and overexpressed or underexpressed in various types of squamous cell carcinomas. Normal PTK expression is relatively restricted and has been analyzed in differentiating epithelial cells of the skin and gastrointestinal tract. PTK6 was also demonstrated to localize in the nuclei of normal and malignant prostate epithelial cells, with a transition to cytoplasmic localization being associated with increased tumor grade. PTK6 fosters anchorage-independent cell growth of NIH3T3; promotes the proliferation of nontransformed mammary epithelial; protects HEK 293 cells from apoptosis; and mediates migration and epidermal growth factor-induced chemotaxis and matrix degradation by a variety of cell types. In contrast, in Rat1a fibroblasts, PTK6 expression does not result in enhanced proliferative properties, but sensitizes the cells to apoptotic stimuli, and PTK6 expression in keratinocyte cell lines promotes the expression of differentiation markers. It was also reported that PTK6 can be induced in normal T lymphocytes after their activation.3 PTK6 expression is developmentally regulated and detected only late in gestation in the mouse, when epithelial linings mature. In mature tissues, PTK6 is expressed in differentiated nondividing cells. PTK6 promotes keratinocyte and epithelial cell differentiation in vivo. PTK6 may participate in regulation of Wnt signaling during development. It was demonstrated that PTK6 directly associates with beta-catenin and that PTK6 expression leads to an inhibition of beta-catenin regulated transcription in vivo and an increase in the levels of TCF4 and the co-repressor TLE/Groucho, which could contribute to growth-inhibitory activities of PTK6 in vivo.4
PTK6 is strongly expressed in a large subset of breast carcinomas, and some metastatic melanomas, moderately overexpressed in some colon cancers, and overexpressed or underexpressed in various types of squamous cell carcinomas. Normal PTK expression is relatively restricted and has been analyzed in differentiating epithelial cells of the skin and gastrointestinal tract. PTK6 was also demonstrated to localize in the nuclei of normal and malignant prostate epithelial cells, with a transition to cytoplasmic localization being associated with increased tumor grade. PTK6 fosters anchorage-independent cell growth of NIH3T3; promotes the proliferation of nontransformed mammary epithelial; protects HEK 293 cells from apoptosis; and mediates migration and epidermal growth factor-induced chemotaxis and matrix degradation by a variety of cell types. In contrast, in Rat1a fibroblasts, PTK6 expression does not result in enhanced proliferative properties, but sensitizes the cells to apoptotic stimuli, and PTK6 expression in keratinocyte cell lines promotes the expression of differentiation markers. It was also reported that PTK6 can be induced in normal T lymphocytes after their activation.3 PTK6 expression is developmentally regulated and detected only late in gestation in the mouse, when epithelial linings mature. In mature tissues, PTK6 is expressed in differentiated nondividing cells. PTK6 promotes keratinocyte and epithelial cell differentiation in vivo. PTK6 may participate in regulation of Wnt signaling during development. It was demonstrated that PTK6 directly associates with beta-catenin and that PTK6 expression leads to an inhibition of beta-catenin regulated transcription in vivo and an increase in the levels of TCF4 and the co-repressor TLE/Groucho, which could contribute to growth-inhibitory activities of PTK6 in vivo.4
REFERENCES
1. Kim, H.L. et al: Biochem. Biophys. Res. Commun. 362:829-34, 2007
2. Kim, H.L. & Lee, S.-T.: J. Biochem. 146:133-9, 2009
3. Kasprzycka, M. et al: Am. J. Path. 168:1631-41, 2006
4. Palka-Hamblin, H.L. et al: J. Cell Sci. 123:236-45, 2010
2. Kim, H.L. & Lee, S.-T.: J. Biochem. 146:133-9, 2009
3. Kasprzycka, M. et al: Am. J. Path. 168:1631-41, 2006
4. Palka-Hamblin, H.L. et al: J. Cell Sci. 123:236-45, 2010
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10209 |
Antigen: | Purified recombinant human PTK6 fragments expressed in E. coli. |
Isotype: | Mouse IgG2b |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
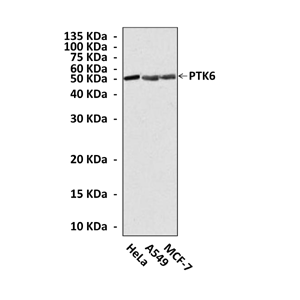
Predicted Molecular Weight of protein: | 52 kDa |
Specificity/Sensitivity: | Detects endogenous PTK6 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
|
| ||||||||||
| ||||||||||
Extended Family Products
|
| ||||||||||
| ||||||||||
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой