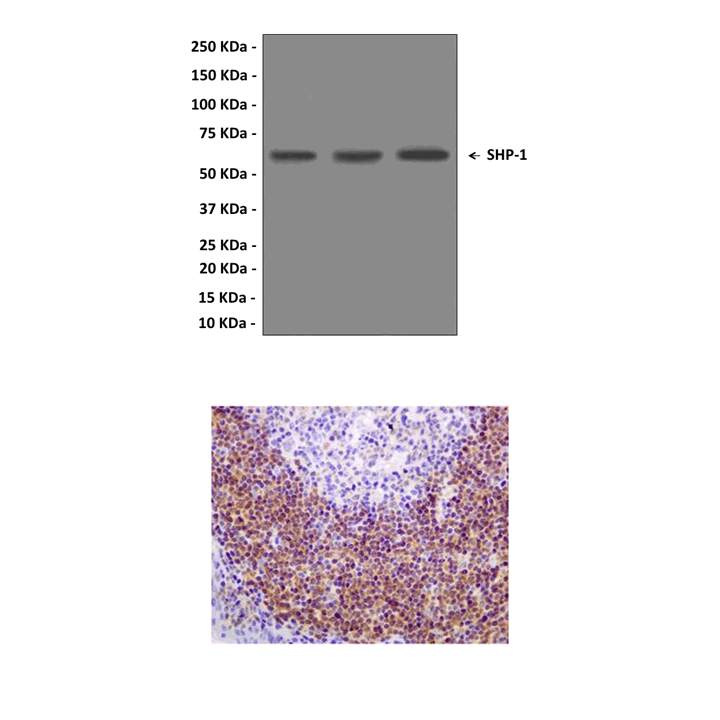
Anti-SHP-1: Rabbit SHP-1 Antibody |
 |
BACKGROUND Two signaling molecules have been implicated in the modulation of immune receptor activation by inhibitory coreceptors: an inositol polyphosphate 5′-phosphatase, SHIP, and a tyrosine phosphatase, SHP-1. Several features have emerged that are common to these inhibitory receptors. A consensus inhibitory motif (ITIM) first identified in Fc-gamma-RIIB, I/LxYxxL, has been recognized in the cytoplasmic domains of this growing family of inhibitory molecules. Inhibitory activity is only seen upon coligation to an ITAM-containing receptor, a class that includes BCR, TCR, and FcRs for IgG and IgE. Phosphorylation of the tyrosine in the ITIM by an ITAM-associated tyrosine kinase is critical to its inhibitory mechanism. In the case of Fc-gamma-RIIB, coligation to the BCR or mast cell Fc-epsilon-RI or Fc-gamma-RIII results primarily in the recruitment of SHIP-1, although in vitro SHP-1 and -2 have also been reported to be associated with this receptor. Similarly, phosphorylation of the ITIM in KIR, CD22, and gp49B1 results in the recruitment of SHP-1, while SHP-2 has been found to be associated with CTLA4.1 Additionally, it was shown that the inhibitory effect of SHP-1 on c-Kit is realized through SHP-1 binding with tyrosine 569 in the c-Kit juxtamembrane domain.2 However, despite these similarities in sequence requirements and potential signaling pathways, the inhibitory responses generated by these receptors are not identical. Indeed, the recruitment of either an inositol polyphosphate 5′-phosphatase (SHIP) or a tyrosine phosphatase is likely to trigger distinct cellular pathways, culminating in inhibitory responses that are phenotypically quite different.
The Src homology 2 domain phosphatase-1 (SHP-1, also called PTP1C, HCP, or SHPTP1) is a tyrosine phosphatase containing two amino-terminal SH2 domains and is expressed primarily by hematopoietic-derived cells. The pivotal role of SHP-1 in the regulation of hemopoietic cell growth and development is now well recognized. In contrast to the structurally similar, ubiquitously expressed SHP-2 (Syp or PTP1D) tyrosine phosphatase, SHP-1 appears to exert primarily inhibitory effects on the signaling cascades in which it participates. SHP-1 has been shown, for example, to suppress the growth-promoting properties of the activated IL-3, Epo, and CSF1 receptors, an effect mediated either directly by receptor dephosphorylation or indirectly by dephosphorylation of receptor-associated protein tyrosine kinases (PTKs). SHP-1 has also been implicated in downregulation of the signaling pathways evoked by engagement of the B- and T-lymphocyte antigen receptors, antigen receptor comodulators such as CD22, Fc RIIB, and CD5, and cytosolic signaling molecules such as Vav and Grb2/Sos1 which are involved in Ras activation.3 The presence of two SH2 domains in SHP-1, as well as the possibility for altering its C-terminal SH2 domain by alternative splicing of a 39-amino-acid segment, provides a structural explanation for the diverse range of molecular interactions in which this phosphotyrosine phosphatase (PTP) appears to participate.4 In addition to the regulation of cell proliferation, SHP-1 has also been implicated in the control of signaling cascades coupling growth factor receptors to hemopoietic cell differentiation.
The Src homology 2 domain phosphatase-1 (SHP-1, also called PTP1C, HCP, or SHPTP1) is a tyrosine phosphatase containing two amino-terminal SH2 domains and is expressed primarily by hematopoietic-derived cells. The pivotal role of SHP-1 in the regulation of hemopoietic cell growth and development is now well recognized. In contrast to the structurally similar, ubiquitously expressed SHP-2 (Syp or PTP1D) tyrosine phosphatase, SHP-1 appears to exert primarily inhibitory effects on the signaling cascades in which it participates. SHP-1 has been shown, for example, to suppress the growth-promoting properties of the activated IL-3, Epo, and CSF1 receptors, an effect mediated either directly by receptor dephosphorylation or indirectly by dephosphorylation of receptor-associated protein tyrosine kinases (PTKs). SHP-1 has also been implicated in downregulation of the signaling pathways evoked by engagement of the B- and T-lymphocyte antigen receptors, antigen receptor comodulators such as CD22, Fc RIIB, and CD5, and cytosolic signaling molecules such as Vav and Grb2/Sos1 which are involved in Ras activation.3 The presence of two SH2 domains in SHP-1, as well as the possibility for altering its C-terminal SH2 domain by alternative splicing of a 39-amino-acid segment, provides a structural explanation for the diverse range of molecular interactions in which this phosphotyrosine phosphatase (PTP) appears to participate.4 In addition to the regulation of cell proliferation, SHP-1 has also been implicated in the control of signaling cascades coupling growth factor receptors to hemopoietic cell differentiation.
REFERENCES
1. Ono, M. et al: Cell 90:293-301, 1997
2. Kozlowski, M. et al: Mol. Cell. Biol. 18:2089-99, 1998
3. Zhang, J. et al: Semin. Immunol. 12:361-78, 2000
4. Poole, A.W. & Jones, M.: Cell Signal. 17:1323-32, 2005
2. Kozlowski, M. et al: Mol. Cell. Biol. 18:2089-99, 1998
3. Zhang, J. et al: Semin. Immunol. 12:361-78, 2000
4. Poole, A.W. & Jones, M.: Cell Signal. 17:1323-32, 2005
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CG1545 |
Antigen: | Synthetic peptide corresponding to the N-terminus of human SHP-1. |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000-1:10000 IP n/d IHC 1:1000-1:2000 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 68 kDa |
Specificity/Sensitivity: | Detects endogenous SHP-1 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой