Anti-SND1: Mouse Staphylococcal Nuclease Domain-Containing Protein 1 Antibody |
 |
BACKGROUND SND1, also known as Tudor-SN and p100, is a highly conserved protein from yeasts to humans. It has been implicated in a variety of cellular processes, such as transcription, processing of edited double-stranded RNA, and splicing regulation. It has five repeated staphylococcal nuclease homology domains and a Tudor-homology domain suggested to act as interaction platforms for nucleic acids and proteins, respectively. SND1 was first identified as a coactivator of the EBV nuclear antigen 2 and was later shown to interact with the oncogene product c-Myb. It is thus a transcriptional coactivator. In addition, Tudor domains have previously been shown to bind methylated ligands, such as methylated lysines and arginines. It has been suggested that the role of Tudor-SN in splicing may involve binding to such methylated ligands or to the methylated 5′ cap of spliceosomal snRNAs.1 A mouse homologue of SND1 was identified as a single-stranded cytosine-rich DNA binding protein, which binds to the d(CTGCC)n sequence derived from mouse minisatellite Pc-1 with high affinity, but not to a complementary guanine-rich repetitive sequence d(GGCAG)n and its double-stranded form. It was recently reported that SND1 is one of the components of the RNA-induced silencing complex (RISC), and disruption of SND1 in C. elegance was shown to cause disruption of small interfering RNA-induced gene silencing. In addition, various studies suggest an involvement of SND1 as a key player in the regulation of gene expression at both transcriptional and posttranscriptional levels through direct binding to RNA in some sequence-specific manner. Along with dicer, SND1 is also the central component of the miR machinery. It was reported that SND1 was up-regulated in androgen independent phenotype of prostate cancer. As the one of main player of miR machinery, SND1 may engage early carcinogenesis, and further androgen independency. Indeed, SND1 may have the potential for identification of the more aggressive and clinically significant prostate cancers.2 Additionally it was also shown that up-regulation of SND1 occurred at a very early stage in colon carcinogenesis and contributed to the posttranscriptional regulation of key players in colon cancer development, including APC and beta-catenin.3
REFERENCES
1. Friberg, A. et al:J. Mol. Biol. 387:921-34, 2009
2. Kuruma, H. et al: Am. J. Pathol. 174:2044-50, 2009
3. Tsuchiya, N. et al: Cancer Res. 67:9569-76, 2007
2. Kuruma, H. et al: Am. J. Pathol. 174:2044-50, 2009
3. Tsuchiya, N. et al: Cancer Res. 67:9569-76, 2007
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10226 |
Antigen: | Purified recombinant human SND1 fragments (aa361-485) expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
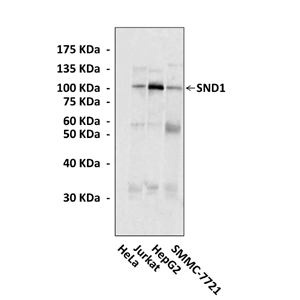
Predicted Molecular Weight of protein: | 102 kDa |
Specificity/Sensitivity: | Detects endogenous SND1 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
|
| ||||||||||
| ||||||||||
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой