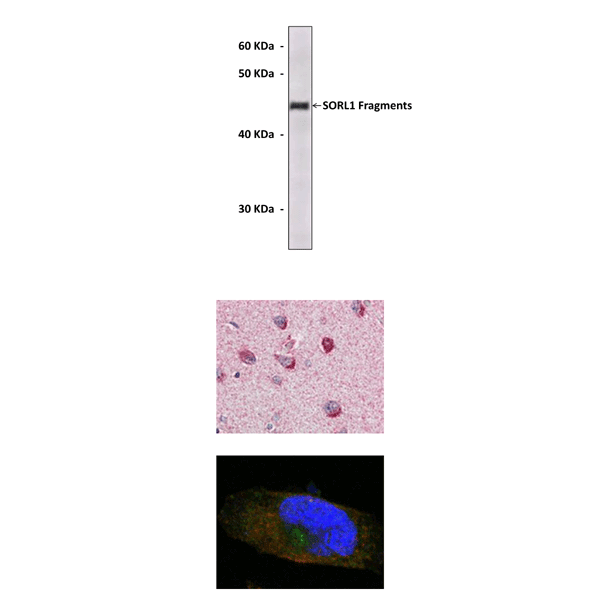
Anti-SORL1: Mouse Neuronal Sortilin-Related Receptor Gene 1 Antibody |
 |
BACKGROUND Neuronal Sortilin-Related Receptor Gene (SORL1, also known as SORLA and LR11) is a susceptibility gene for late-onset Alzheimer's disease (AD). It is located on chromosome 11q23.2-q24.2 and encodes a 250-kD membrane protein expressed in neurons of the central and peripheral nervous system. It is known to be involved in intracellular trafficking between the membrane and intracellular organelles, interacting with APP in endosomes and the trans-Golgi network (TGN) in both in vitro and in vivo experiments. The current data suggest that underexpression of SORL1 leads to overexpression of amyloid beta (Abeta), which has been associated with a higher risk of developing AD.1 The accumulation of Abeta peptide, a neurotoxic proteolytic derivative of the amyloid precursor protein (APP) is a central event in the pathogenesis of AD. Thus, accumulation of Abeta in the brain is associated with disease-causing inherited variants in the amyloid precursor protein (APP), presenilin 1 (PS1) presenilin 2 (PS2) and apolipoprotein E (APOE) genes. The generation of Abeta occurs in several subcellular compartments, but a principle location is during the re-entry and recycling of APP from the cell surface via the endocytic pathway. The inherited variants in these pathways might modulate APP processing, and thereby might affect risk for AD. SORL1 plays a key physiological role in the differential sorting of the amyloid precursor protein (APP) holoprotein. In the presence of SORL1, APP holoprotein is recovered via the retromer. In the absence of SORL1, APP is released into late endosomal pathways where it is subjected to beta- and subsequently γ-secretase cleavage that generate Abeta.2
APP holoprotein is synthesized in the endoplasmic reticulum (ER) and Golgi. Proteolytic cleavage through the Abeta peptide domain by ADAM17 and other α-secretase enzymes generates N-terminal soluble APPsalpha and membrane-bound APP-CTFα fragments. Sequential cleavage by BACE1 (beta-secretase) generates N-terminal APPsbeta and membrane bound APP-CTFbeta fragments. The latter undergoes presenilin-dependent gamma-secretase cleavage to generate Abeta and amyloid intracellular domain (AICD). SORL1 binds both APP holoprotein and VPS35 and acts as a sorting receptor for APP holoprotein. Absence of SORL1 switches APP holoprotein away from the retromer recycling pathway, and instead directs APP into the beta-secretase cleavage pathway, increasing APPsbeta production and then into the gamma-secretase cleavage pathway to generate Abeta. Blockade of the retromer complex (RC) by inhibiting retromer complex proteins such as VPS26 or VPS35 has a similar effect, also increasing APPsbeta and Abeta production.3
APP holoprotein is synthesized in the endoplasmic reticulum (ER) and Golgi. Proteolytic cleavage through the Abeta peptide domain by ADAM17 and other α-secretase enzymes generates N-terminal soluble APPsalpha and membrane-bound APP-CTFα fragments. Sequential cleavage by BACE1 (beta-secretase) generates N-terminal APPsbeta and membrane bound APP-CTFbeta fragments. The latter undergoes presenilin-dependent gamma-secretase cleavage to generate Abeta and amyloid intracellular domain (AICD). SORL1 binds both APP holoprotein and VPS35 and acts as a sorting receptor for APP holoprotein. Absence of SORL1 switches APP holoprotein away from the retromer recycling pathway, and instead directs APP into the beta-secretase cleavage pathway, increasing APPsbeta production and then into the gamma-secretase cleavage pathway to generate Abeta. Blockade of the retromer complex (RC) by inhibiting retromer complex proteins such as VPS26 or VPS35 has a similar effect, also increasing APPsbeta and Abeta production.3
REFERENCES
1. Lee, J.H. et al: Curr Neurol Neurosci Rep. 8:384-91, 2008
2. Rogaeva, E. et al: Nature Genet. 39:168-77, 2007
3. Ma, Q. et al: Arch. Neurol. 64:448-57, 2009
2. Rogaeva, E. et al: Nature Genet. 39:168-77, 2007
3. Ma, Q. et al: Arch. Neurol. 64:448-57, 2009
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10228 |
Antigen: | Purified recombinant human SORL1 fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:200 ICC 1:200 FACS n/d |
Predicted Molecular Weight of protein: | 250 kDa |
Specificity/Sensitivity: | Detects endogenous SORL1 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой