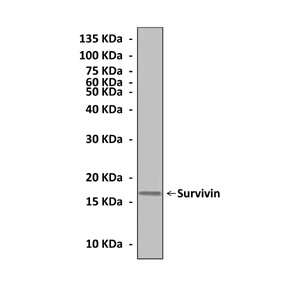
Anti-Survivin: Mouse Survivin Antibody |
 |
BACKGROUND Survivin, a novel human number of the inhibitor of apoptosis protein (IAP) family, has been suggested to directly inhibit caspase-3 and -7 activity or conjugate caspase-9, and regulate the G2/M phase by interact with spindle microtubules. Survivin is expressed in fetal development, in normal endometrium primarily during the secretory phase of the menstrual cycle and in cancers, whereas no transcripts were detected in terminally differentiated adult tissues. Expression of Survivin on breast, neuroblastoma, lung, esophageal, and colorectal cancers correlates with an unfavorable prognosis, which is shortened survival and/or a shortened time to recurrence.1 IAP family members usually contain multiple baculovirus IAP repeat (BIR) domains, but Survivin has only a single BIR domain. It also lacks a C-terminus RING finger domain. At least four transcript variants encoding distinct isoforms have been found for Aurvivin.
Besides its role as an IAP, Survivin acts as a subunit of the chromosomal passenger complex (CPC) and as a regulator of microtubule dynamics. The CPC, is composed of the Aurora-B kinase, Borealin, and INCENP, corrects attachment errors between chromosomes and the mitotic spindle, regulates the quality-control checkpoint, and ensures the correct completion of cytokinesis. The typical chromosomal passenger localization pattern of Survivin can be observed not only in normal but also in tumor cells. It was shown that Borealin is complexed with Survivin, which can bind to Aurora-B kinase and is incorporated into the CP holocomplex by interacting with INCENP. The NES in Survivin mediates recruitment of Crm1/RanGTP. The complex with Crm1/RanGTP plays a role in mitotic spindle formation by serving as a physical scaffold to help deliver the Ran effector molecule TPX2 to microtubules. This process might be catalyzed by the activity of the guanine nucleotide exchange factor RCC1 or TD60. Hydrolysis of RanGTP, by factors like RanBPs/Ran-GAP1, may facilitate the release of Crm1 and deposition of the CPC at the inner centromere.2
In normal cells, expression of Survivin peaks at the G2/M transition of the cell cycle, whereas the cell cycle–dependent transcriptional control of Survivin is deregulated by various oncogenic pathways in cancer cells. Hence, Survivin is found in the majority of interphase tumor cells in patients, which supports its bifunctional role. Moreover, the observation that Survivin can be found not only in the cytoplasm but also in the nucleus of tumor and proliferating normal cells (e.g., endothelial and CD34+ stem cells) stimulated the hypothesis that these subcellular pools may coincide with different Survivin functions. Nuclear Survivin was suspected to control cell division, whereas cytoplasmic/mitochondrial Survivin was considered cytoprotective. A failure in passing on the duplicated genetic material to both daughter cells together with resistance to apoptosis can contribute to cellular transformation and cancer progression. Survivin seems to intersect both processes.3
REFERENCES
1. Altieri, D.C.: Trends Mol. Med. 7:542-7, 2001
2. Xia, F. et al: Mol. Cell. Biol. 28:5299-5311, 2008
3. Stauber, R.H. et al: Cancer Res. 67:5999-6002, 2007
2. Xia, F. et al: Mol. Cell. Biol. 28:5299-5311, 2008
3. Stauber, R.H. et al: Cancer Res. 67:5999-6002, 2007
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10232 |
Antigen: | Purified recombinant human surviving proteins expressed in E. coli. |
Isotype: | Mouse IgG |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 16 kDa |
Specificity/Sensitivity: | Detects endogenous Survivin proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой