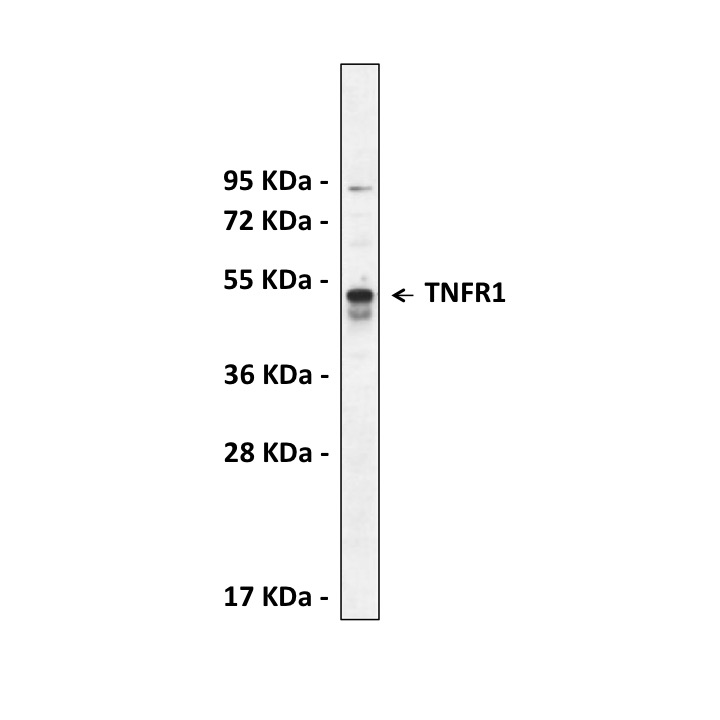
Anti-TNFR1: Rabbit Tumor Necrosis Factor Receptor 1 Antibody |
 |
BACKGROUND Tumor Necrosis Factor (TNF) is a highly pleiotropic cytokine that elicits diverse cellular responses ranging from proliferation and differentiation to activation of apoptosis. The different biological activities are mediated by two distinct cell surface receptors: 55 kDa TNF receptor 1 (TNFR-1, p55, CD120a) or 75 kDa TNF-R2 (p75, CD120b). TNFR-1 is widely expressed on a number of cell types. TNFR-2 is found predominantly on leukocytes and endothelial cells and is encoded on chromosome 1p. Both TNFR1 and TNFR2 contain repeating cysteine-rich extracellular motifs. TNF-R1 appears to be the key mediator of TNF signaling. TNF homotrimers induce signaling by aggregation of membrane receptors. TNFR1 contains a cytoplasmic death domain (DD) characteristic of many members of the TNF superfamily.1 Upon binding of its ligand, TNF-R1 permits the assembly of the TNFR1 signaling complex through the dissociation of silencer of death domains (SODD) and subsequent binding of TNF receptor associated death domain (TRADD). Binding of TRADD then allows the recruitment of other adaptor proteins including receptor interacting protein (RIP) and TNF receptor associated factor 2 (TRAF2). This complex then leads to RIP-dependent activation of NFκB signaling to initiate pro-survival signaling, cellular proliferation, and cytokine production. This membrane associated complex of ligand-engaged TNFR1 with TRADD, TRAF2, and RIP also recruits cellular inhibitor of apoptosis proteins 1 and 2 (cIAP 1,2) resulting in activation of ERK, JNK, p38 MAP kinase, and ceramide/sphingomyelinase pathways. The kinetics of JNK activation is particularly important in determining the functional outcome of a TNF signal. Acute, transient TNF-induced JNK activation, which is cytoprotective, results from TAK1-dependent phosphorylation, whereas sustained JNK activation leading to caspase-dependent apoptosis depends upon JNK phosphorylation by apoptosis signal-regulating kinase 1 (ASK1). Knock-in mice expressing mutant TNF that is no longer a substrate for TACE cleavage (and therefore remains membrane bound) have demonstrated solTNF to be the primary mediator of TNF-dependent inflammatory responses. In these transgenic mice, lymphoid organ development is relatively normal (with the exception of primary B cell follicles), but the initiation of autoimmune pathology is severely compromised.2
In addition to TNFR1-induced activation of stress-induced kinase signaling in contributing to apoptotic signaling, TNFR1 can be internalized after TNF binding and this leads to dissociation of the TRADD/TRAF2/RIP complex and association of Fas-associated DD (FADD), recruitment of pro-caspase 8, and formation of the death-inducing signaling complex (DISC), triggering activation of the executioner caspases through the extrinsic apoptosis pathway. Caspase-8, combined with its ability to induce apoptosis through the extrinsic pathway, also triggers the intrinsic apoptosis pathway by cleaving the pro-apoptotic Bcl-2 family members Bax and Bid to initiate mitochondrial-induced apoptosis. The complexity of TNFR1-mediated signaling is what allows many divergent (ie, proliferation, activation, apoptosis) outcomes to occur as a result of TNF signaling.3
TNF actions may also be constrained in part by metalloprotease-dependent cleavage of membrane-bound TNF receptors, which can be triggered by a number of proinflammatory stimuli. Cleavage is thought to contribute to the clearance of TNFR1 and TNFR2 from the membrane, and produces a pool of soluble receptors that may attenuate the inflammatory response by competing with membrane-bound receptors.4 Mice homozygous for a targeted mutation in an enzyme that cleaves several membrane proteins (including TNF and TNFR2) have numerous developmental defects, emphasizing the physiologic importance of ectodomain shedding.
In addition to TNFR1-induced activation of stress-induced kinase signaling in contributing to apoptotic signaling, TNFR1 can be internalized after TNF binding and this leads to dissociation of the TRADD/TRAF2/RIP complex and association of Fas-associated DD (FADD), recruitment of pro-caspase 8, and formation of the death-inducing signaling complex (DISC), triggering activation of the executioner caspases through the extrinsic apoptosis pathway. Caspase-8, combined with its ability to induce apoptosis through the extrinsic pathway, also triggers the intrinsic apoptosis pathway by cleaving the pro-apoptotic Bcl-2 family members Bax and Bid to initiate mitochondrial-induced apoptosis. The complexity of TNFR1-mediated signaling is what allows many divergent (ie, proliferation, activation, apoptosis) outcomes to occur as a result of TNF signaling.3
TNF actions may also be constrained in part by metalloprotease-dependent cleavage of membrane-bound TNF receptors, which can be triggered by a number of proinflammatory stimuli. Cleavage is thought to contribute to the clearance of TNFR1 and TNFR2 from the membrane, and produces a pool of soluble receptors that may attenuate the inflammatory response by competing with membrane-bound receptors.4 Mice homozygous for a targeted mutation in an enzyme that cleaves several membrane proteins (including TNF and TNFR2) have numerous developmental defects, emphasizing the physiologic importance of ectodomain shedding.
REFERENCES
1. Watts, T.H.: Ann. Rev. Immunol. 23:23-68, 2005
2. Michael Karin, M. & Gallaghe, E.: Immunol. Rev. 228:225-40, 2009
3. Aggarwal, B.B.: Nature Rev. Immunol. 3:745-56, 2003
4. van Mierlo, G.J.D. et al: J. Immunol. 180:2747-51, 2008
2. Michael Karin, M. & Gallaghe, E.: Immunol. Rev. 228:225-40, 2009
3. Aggarwal, B.B.: Nature Rev. Immunol. 3:745-56, 2003
4. van Mierlo, G.J.D. et al: J. Immunol. 180:2747-51, 2008
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CG1602 |
Antigen: | Synthetic TNFR1 peptide |
Isotype: | Rabbit IgG |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC n/d ICC 1:10-1:50 FACS n/d |
Predicted Molecular Weight of protein: | 50495 Da |
Specificity/Sensitivity: | Detects endogenous TNFR1 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой