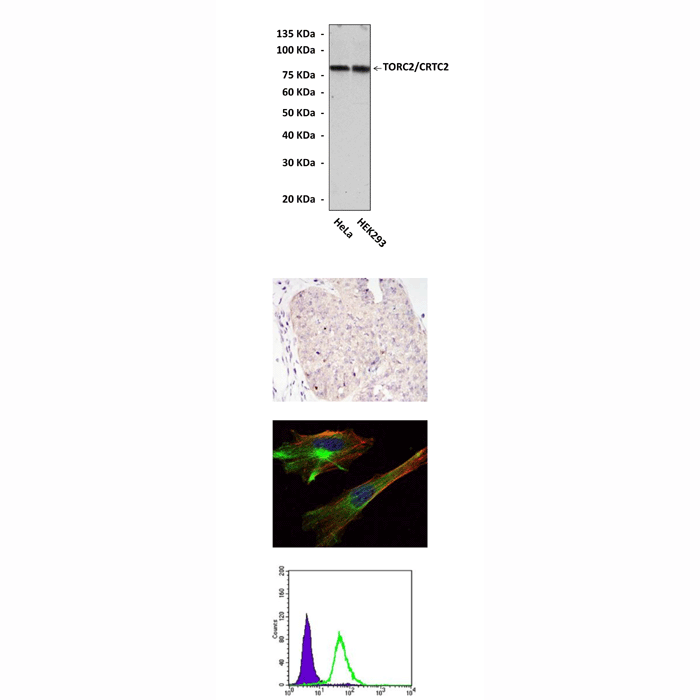
Anti-TORC2/CRTC2: Mouse TORC2/CRTC2 Antibody |
 |
BACKGROUND Transducer of Regulated CREB (TORC) 1, 2, and 3 were identified from a lymphocyte cDNA library as a family of CREB co-activators that bind to CREB and enhance CRE-mediated transcription in an Ser-133 phosphorylation-independent manner. It was reported that TORCs are activated by the calcium-regulated phosphatase, calcineurin. Dephosphorylation of TORC by the phosphatase triggers release from 14-3-3 proteins and translocation from cytoplasm to nucleus.1 TORC2 (also known as CRTC2) is a major transcriptional coregulator for hepatic glucose output in response to fasting in mammals. Glucose homeostasis is regulated systemically by hormones such as insulin and glucagon, and at the cellular level by energy status. Glucagon enhances glucose output from the liver during fasting by stimulating the transcription of gluconeogenic genes via the cyclic AMP-inducible factor CREB (CRE binding protein). When cellular ATP levels are low, however, the energy-sensing kinase AMPK inhibits hepatic gluconeogenesis. Signals that activate AMPK attenuate the gluconeogenic programme by promoting TORC2 phosphorylation and blocking its nuclear accumulation.2 During feeding, increases in circulating pancreatic insulin inhibit hepatic glucose output through the activation of the Ser/Thr kinase AKT and subsequent phosphorylation of the forkhead transcription factor FOXO1. Under fasting conditions, FOXO1 increases gluconeogenic gene expression in concert with the cAMP responsive coactivator TORC2. In response to pancreatic glucagon, TORC2 is de-phosphorylated at Ser 171 and transported to the nucleus, in which it stimulates the gluconeogenic programme by binding to CREB. Insulin inhibits gluconeogenic gene expression during re-feeding by promoting the phosphorylation and ubiquitin-dependent degradation of TORC2. Insulin disrupts TORC2 activity by induction of the Ser/Thr kinase SIK2, which has shown to undergoes AKT2-mediated phosphorylation at Ser 358. Activated SIK2 in turn stimulated the Ser 171 phosphorylation and cytoplasmic translocation of TORC2. Phosphorylated TORC2 was degraded by the 26S proteasome during re-feeding through an association with COP1, a substrate receptor for an E3 ligase complex that promoted TORC2 ubiquitination at Lys 628. Because TORC2 protein levels and activity were increased in diabetes owing to a block in TORC2 phosphorylation.3 The signaling pathway involves the kinase SIK2 and the E3 ligase COP1. The findings point to TORC2 and SIK2 as potential therapeutic targets in type II diabetes. Individuals with type 2 diabetes often exhibit fasting hyperglycaemia due to elevated gluconeogenesis; compounds that enhance TORC2 phosphorylation may offer therapeutic benefits in this setting.4
REFERENCES
1. Ryu, D. et al: Cell Metab. 9:240-51, 2009
2. Koo, S.-H. et al: Nature 437:1109-11, 2005
3. Dentin, R. et al: Nature 449:366-9, 2007
4. Muraoka, M. et al: Am. J. Physiol. Endocrin. Metab. 296:E1340-9, 2009
2. Koo, S.-H. et al: Nature 437:1109-11, 2005
3. Dentin, R. et al: Nature 449:366-9, 2007
4. Muraoka, M. et al: Am. J. Physiol. Endocrin. Metab. 296:E1340-9, 2009
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10299 |
Antigen: | Raised against recombinant human TORC2 fragments expressed in E. coli. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human, Mouse, Rat |
Applications & Suggested starting dilutions:* | WB 1:1000 IP n/d IHC 1:50 - 1:200 ICC 1:50 - 1:200 FACS 1:50 - 1:200 |
Predicted Molecular Weight of protein: | 80 kDa |
Specificity/Sensitivity: | Detects endogenous TORC2 proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой