Anti-TrkC: Mouse TrkC Antibody |
 |
BACKGROUND Trk (neurotrophin) receptors are single transmembrane catalytic receptors with intracellular tyrosine kinase activity. Trk receptors are coupled to the Ras, Cdc42/Rac/RhoG, MAPK, PI 3-K and PLCgamma signaling pathways. There are four members of the Trk family; TrkA, TrkB and TrkC and a related p75NTR receptor. p75NTR lacks tyrosine kinase activity and signals via NF-kappaB activation. Each family member binds different neurotrophins with varying affinities. TrkA potently binds nerve growth factor (NGF) and is involved in differentiation and survival of neurons and in control of gene expression of enzymes involved in neurotransmitter synthesis. TrkB has highest affinity for brain-derived neurotrophic factor (BDNF) and is involved in neuronal plasticity, longterm potentiation and apoptosis of CNS neurons. TrkC is activated by neurotrophin-3 (NT-3) and is found on proprioceptive sensory neurons. p75NTR binds neurotrophin precursors with high affinity and retains low affinity to the mature cleaved forms. TrkA was originally identified as an oncogene as it is commonly mutated in cancers, particularly colon and thyroid carcinomas.
The transmembrane protein-tyrosine kinase TrkC is the high affinity receptor for neurotrophin-3 (NT-3). The interaction between TrkC and NT-3 elicits a more efficient biological response than when NT-3 binds to its other Trk receptors. Thus, TrkC may play an important role in mediating the neurotrophic effects of NT-3.1 Upon neurotrophin binding, TrkC become dimerized and activated. It phosphorylates itself. The putative regulatory tyrosines 674, 678, and 679 in the tyrosine kinase homology region are major ligand-stimulated autophosphorylation sites, which are required for full-activation of TrkC tyrosine kinase activity. Moreover, it was shown that the putative phospholipase Cgamma and Shc binding sites on TrkC (tyrosines 485 and 789, respectively) are indeed autophosphorylated. Kinase inserts occur between subdomains VII and VIII of the tyrosine kinase homology region and immediately C-terminal to tyrosines 674, 678, and 679, and specifically block the autophosphorylation of tyrosine 785 and this prevents phospholipase Cgamma association with TrkC and its tyrosine phosphorylation.2 TrkC signaling is mediated by downstream MAPK and PI-3 kinase pathway. Signaling through this kinase leads to cell differentiation and may play a role in the development of proprioceptive neurons that sense body position. Moreover, it was shown that neurotrophin 3 activation of TrkC induces Schwann cell migration through the Rho GTPases (Rac1 and Cdc42) and JNK signaling pathway.3 In addition, TrkC may atc outside of neural system. It was demonstrated that TrkC directly binds to the BMP type II receptor (BMPRII) in colon cancer cells, thereby preventing it from interacting with the BMPRI. This activity requires a functional TrkC protein tyrosine kinase, and the BMPRII seems to be a direct target of TrkC. TrkC can block BMP tumor-suppressor activity.4 Mutations in TrkC have been associated with medulloblastomas, secretory breast carcinomas and other cancers.
The transmembrane protein-tyrosine kinase TrkC is the high affinity receptor for neurotrophin-3 (NT-3). The interaction between TrkC and NT-3 elicits a more efficient biological response than when NT-3 binds to its other Trk receptors. Thus, TrkC may play an important role in mediating the neurotrophic effects of NT-3.1 Upon neurotrophin binding, TrkC become dimerized and activated. It phosphorylates itself. The putative regulatory tyrosines 674, 678, and 679 in the tyrosine kinase homology region are major ligand-stimulated autophosphorylation sites, which are required for full-activation of TrkC tyrosine kinase activity. Moreover, it was shown that the putative phospholipase Cgamma and Shc binding sites on TrkC (tyrosines 485 and 789, respectively) are indeed autophosphorylated. Kinase inserts occur between subdomains VII and VIII of the tyrosine kinase homology region and immediately C-terminal to tyrosines 674, 678, and 679, and specifically block the autophosphorylation of tyrosine 785 and this prevents phospholipase Cgamma association with TrkC and its tyrosine phosphorylation.2 TrkC signaling is mediated by downstream MAPK and PI-3 kinase pathway. Signaling through this kinase leads to cell differentiation and may play a role in the development of proprioceptive neurons that sense body position. Moreover, it was shown that neurotrophin 3 activation of TrkC induces Schwann cell migration through the Rho GTPases (Rac1 and Cdc42) and JNK signaling pathway.3 In addition, TrkC may atc outside of neural system. It was demonstrated that TrkC directly binds to the BMP type II receptor (BMPRII) in colon cancer cells, thereby preventing it from interacting with the BMPRI. This activity requires a functional TrkC protein tyrosine kinase, and the BMPRII seems to be a direct target of TrkC. TrkC can block BMP tumor-suppressor activity.4 Mutations in TrkC have been associated with medulloblastomas, secretory breast carcinomas and other cancers.
REFERENCES
1. Lamballe, F. et al: Cell 66:967-979, 1991
2. Guiton, M. et al: J. Biol. Chem. 270:20384-90, 1995
3. Yamauchi, J. et al: Proc. Natl. Acad. Sci. USA 100:14421-6, 2003
4. Jin, W. et al: Cancer Res. 67:9869-77, 2007
2. Guiton, M. et al: J. Biol. Chem. 270:20384-90, 1995
3. Yamauchi, J. et al: Proc. Natl. Acad. Sci. USA 100:14421-6, 2003
4. Jin, W. et al: Cancer Res. 67:9869-77, 2007
Products are for research use only. They are not intended for human, animal, or diagnostic applications.
Параметры
Cat.No.: | CP10188 |
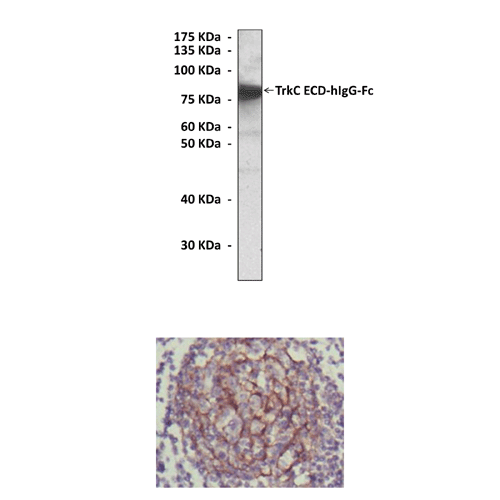
Antigen: | Purified recombinant human TrkC ECD (extracellular domain) expressed in 293 cells. |
Isotype: | Mouse IgG1 |
Species & predicted species cross- reactivity ( ): | Human |
Applications & Suggested starting dilutions:* | WB 1:1000 IP 1:50 IHC 1:100 ICC n/d FACS n/d |
Predicted Molecular Weight of protein: | 145 kDa |
Specificity/Sensitivity: | Detects TrkC proteins without cross-reactivity with other family members. |
Storage: | Store at -20°C, 4°C for frequent use. Avoid repeated freeze-thaw cycles. |
*Optimal working dilutions must be determined by end user.
Документы
Информация представлена исключительно в ознакомительных целях и ни при каких условиях не является публичной офертой